Media Contacts
Brookhaven National Laboratory
Media & Communications Office
P.O. Box 5000 — Bldg. 400C
Upton, NY 11973
Phone: (631) 344-2345
-

Peter Genzer
Manager , Media & Communications Office; Deputy Director
(631) 344-3174, genzer@bnl.gov
-

-

75th Anniversary Celebration, Q&A
January 27, 2022
To kick off the Brookhaven Lab's 75th anniversary year, U.S. Energy Secretary Jennifer Granholm, Lab leaders, staff, and members of the public gathered for a live, virtual event on Jan. 27. Secretary Granholm offered Brookhaven her congratulations and thanks. Then three Lab leaders shared their vision for the future of nuclear and particle physics, climate science, quantum information science, and more before taking questions from the audience.

High-resolution Lidar Sees Birth Zone of Cloud Droplets
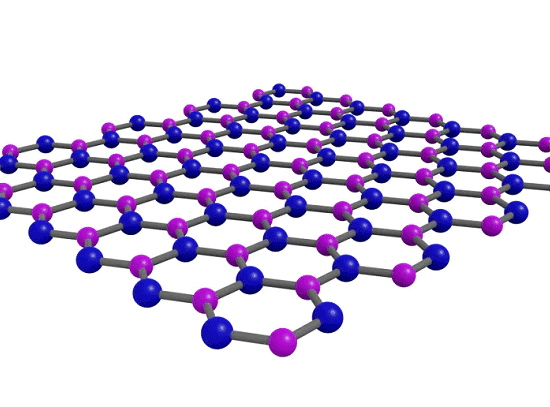
Perfecting the View on a Crystal's Imperfection

Theoretical Physicist Dekrayat Almaalol Receives 2024 Leona Woods Lectureship Award

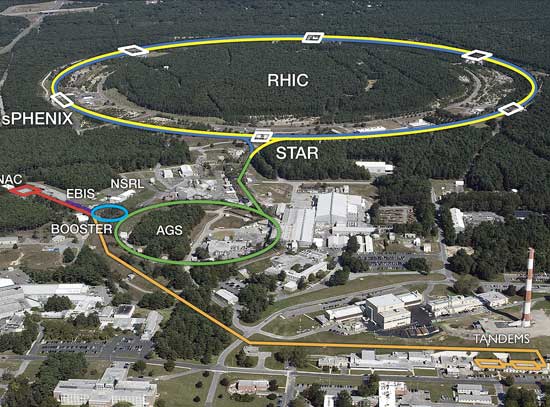
Governor Hochul Launches $100 Million Investment in Brookhaven National Lab for Transformative Electron-Ion Collider Project

SLAC Completes Construction of the Largest Digital Camera Ever Built for Astronomy
-
Q&A: Cosmic rays, space weather and larger questions about the universe
-
US Electron-Ion Collider hits construction milestone
-
Brookhaven National Laboratory's Meng Xie helps boost biomass in sorghum
-
Brookhaven National Lab's $3B supercollider gets major state funding
-
NYS is investing $100M in Brookhaven Lab's electron-ion collider
- Newsclips are articles in the press that are either about Brookhaven or are of interest to the Brookhaven community. Posting of these articles does not imply an endorsement of their content.