Cocaine's Effects on Brain Metabolism May Contribute to Abuse
Study in mice shows drug's effects extend beyond dopamine, brain's "reward" chemical
February 18, 2008
UPTON, NY - Many studies on cocaine addiction - and attempts to block its addictiveness - have focused on dopamine transporters, proteins that reabsorb the brain's "reward" chemical once its signal is sent. Since cocaine blocks dopamine transporters from doing their recycling job, it leaves the feel-good chemical around to keep sending the pleasure signal. Now a new study conducted at the U.S. Department of Energy's Brookhaven National Laboratory suggests that cocaine's effects go beyond the dopamine system. In the study, cocaine had significant effects on brain metabolism, even in mice that lack the gene for dopamine transporters.
"In dopamine-transporter-deficient mice, these effects on metabolism are clearly independent of cocaine's effects on dopamine," said Brookhaven neuroscientist Panayotis (Peter) Thanos, who led the research. "These metabolic factors may be a strong regulator of cocaine use and abuse, and may also suggest new avenues for addiction treatments." The study will appear in the May 2008 issue of the journal Synapse, and will be available online on Monday, February 18, 2008.
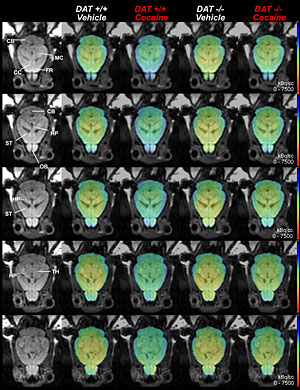 enlarge
enlarge
Magnetic resonance imaging (MRI) and positron emission tomography (PET) scans showing the effect of cocaine on brain metabolism in mice with normal levels of dopamine transporter proteins (DAT +/+) and mice lacking dopamine transporters (DAT -/-). Cocaine was compared with saline treatment (vehicle). Cocaine use blunted whole brain metabolism in both groups of mice (indicated by a reduced amount of yellow visible on the cocaine images), and had a particularly significant effect on the thalamus (TH) in DAT -/- mice. These results indicate that cocaine affects the brain in ways not modulated by its blockade of dopamine transporter proteins.
The scientists used positron emission tomography, or PET scanning, to measure brain metabolism in dopamine-transporter deficient mice (known as DAT knockouts) and in littermates that had normal dopamine transporter levels. In this technique, the scientists administer a radioactively labeled form of sugar (glucose) - the brain's main "fuel" - and use the PET scanner to track its site-specific concentrations in various brain regions. They tested the mice before and after cocaine administration, and compared the results to mice treated with saline instead of the drug.
Before any treatment, mice lacking dopamine transporters had significantly higher metabolism in the thalamus and cerebellum compared with normal mice. This elevated metabolism may be linked to chronically high levels of dopamine in the DAT knockout mice. It also suggests that dopamine levels may play an important role in modulating glucose levels in these brain areas, which play important roles integrating sensory information, learning, and motor function.
Interestingly, DAT knockout mice have been suggested as an animal model for attention-deficit hyperactivity disorder (ADHD). Elevated metabolism due to persistent elevated dopamine levels may be a factor contributing to the symptoms of ADHD, Thanos said.
After the scientists administered cocaine, whole brain metabolism decreased in both groups of mice, but more significantly in normal mice than in DAT knockouts. The scientists were able to detect this reduction in metabolism in a wide range of brain regions in the normal mice, suggesting that these decreases in metabolism are somehow associated with the blockade of dopamine transporters by cocaine.
The scientists also observed a reduction in metabolism in the thalamus region in the DAT knockout mice. This effect may likely be due to the effect of cocaine on other neurotransmitter systems, for example, norepinepherine or serotonin.
In summary, cocaine exposure has an effect on regional brain activity, which is mostly driven by dopamine action and to a secondary degree norepinephrine or serotonin. These results also support the idea that the thalamus and the cerebellum play key roles in cocaine's mechanism of effect on sensory input, learning, and motor function. This is particularly of interest in better understanding the mechanism of cocaine addiction as well as the neurobiology of ADHD.
The research was funded by The National Institute on Alcohol Abuse and Alcoholism Intramural Research Program at the National Institutes of Health and by the Office of Biological and Environmental Research within the U.S. Department of Energy's (DOE) Office of Science. DOE has a long-standing interest in research on brain chemistry gained through brain-imaging studies. Brain-imaging techniques such as PET are a direct outgrowth of DOE's support of basic physics and chemistry research.
All research involving laboratory animals at Brookhaven National Laboratory is conducted under the jurisdiction of the Lab's Institutional Animal Care and Use Committee in compliance with the Public Heath Service (PHS) Policy on Humane Care and Use of Laboratory Animals, the U.S. Department of Agriculture's Animal Welfare Act, and the National Academy of Sciences' Guide for the Care and Use of Laboratory Animals. This research has enhanced understanding of a wide array of human medical conditions including cancer, drug addiction, Alzheimer's and Parkinson's diseases, and normal aging and has led to the development of several promising treatment strategies.
For further information on Peter Thanos' lab and research visit http://www.bnl.gov/thanoslab.
2008-10746 | INT/EXT | Newsroom









