Accelerating a Key Reaction in Fuel Cells and Batteries
May 31, 2017
 enlarge
enlarge
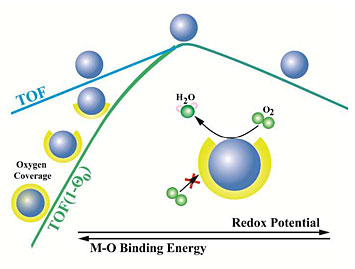
The turnover frequency (TOF) of oxygen-reduction reaction (ORR) catalysts was shown to be limited by the blockage of active sites by reaction intermediates for those catalysts that have low redox potentials.
The Science
The oxygen-reduction reaction is one of the most important and fundamental chemical reactions that takes place within energy conversion systems, such as fuel cells and certain battery types. During the reaction, which occurs at the fuel cell cathode (its positive terminal), the O2 molecule is cleaved in a multi-step process. The reaction is very sluggish on its own and thus is aided by catalysts, often metals or metal alloys. However, scientists still don’t have a full grasp on the limitations of these catalysts and, therefore, how to improve them. In this work, researchers pinpointed some of the factors that affect the catalysts’ performance, a first step toward determining how to improve them.
The Impact
The oxygen-reduction reaction is necessary to the performance of fuel cells and some batteries. It is sped up by catalysts such as platinum, which is the most efficient of the metal element catalysts but is also expensive and not particularly abundant. Platinum alloys (with metals such as cobalt, nickel, and copper) and non-platinum catalysts have been used with success, even showing greater activity. A more efficient catalyst will improve the effectiveness and longevity of energy-storage devices, such as fuel cells, allowing for less or even zero platinum to be used.
Research Summary
Scientists gauged the performance of platinum (Pt) and non-Pt catalysts for the oxygen-reduction reaction (ORR) by taking electrochemical measurements and via x-ray absorption spectroscopy, which can return information on a sample’s chemical and electronic structures based on how the sample absorbs (or scatters) x-rays. The x-ray studies were done at the National Synchrotron Light Source II’s Inner-Shell Spectroscopy (ISS) beamline and at the Argonne National Laboratory’s Advanced Photon Source beamline 10-ID.
The research focused on the catalysts’ limitations as a result of two factors: first, the redox potential, a measure (in volts) of a catalyst’s tendency to gain electrons; second, the degree to whether the catalyst has blocked “active sites,” which are the atomic-scale regions on the catalyst surface where the reaction, or part of it, takes place. These sites can be blocked by reaction intermediates, which are compounds that are created partway through the multi-step reaction. In other words, molecules created during the reaction that the catalyst accelerates can partially inactivate that catalyst.
The site-blocking effect was investigated on both Pt-based and non-Pt catalysts. The group observed that the role of the site-blocking effect on the ORR varies drastically depending on the redox potential of the catalyst active sites. ORR catalysts with overly high redox potentials were limited not by active-site blocking but by their own inherent activity (the number of catalytic cycles occurring at the site per unit time), whereas those with low redox potentials were limited by either their inherent activity or the site blocking effect – or a combination of both, depending on the redox potential. The researchers conclude that the role of site blocking has been greatly underestimated in some cases.
Download research summary slide
Contact
Qingying Jia
Northeastern University
q.jia@neu.edu
Publications
J. Li et al., “Asymmetric Volcano Trend in Oxygen Reduction Activity of Pt and non-Pt Catalysts: In Situ Identification of the Site-Blocking Effect.” J. Am. Chem. Soc. 139, 1384-1387 (2017) DOI: 10.1021/jacs.6b11072
Funding
The authors appreciate financial assistance from the U.S. Department of Energy (DOE), EERE (DE-EE-0000459). This work was also supported by the Natural Science Foundation of China (21336003, 21476138). MRCAT operations are supported by the Department of Energy and the MRCAT member institution. This research used resources of the Advanced Photon Source, a DOE Office of Science User Facility operated for the DOE Office of Science by Argonne National Laboratory under Contract No. DEAC02-06CH11357. Part of this research used ISS 8-ID beamline of the National Synchrotron Light Source II, a DOE Office of Science User Facility operated for the DOE Office of Science by Brookhaven National Laboratory under Contract No. DE-AC02-98CH10886 and DE-SC0012704.
2017-13095 | INT/EXT | Newsroom









