X-rays Reveal Unexpected Protein Function in Plants
November 28, 2023
 enlarge
enlarge
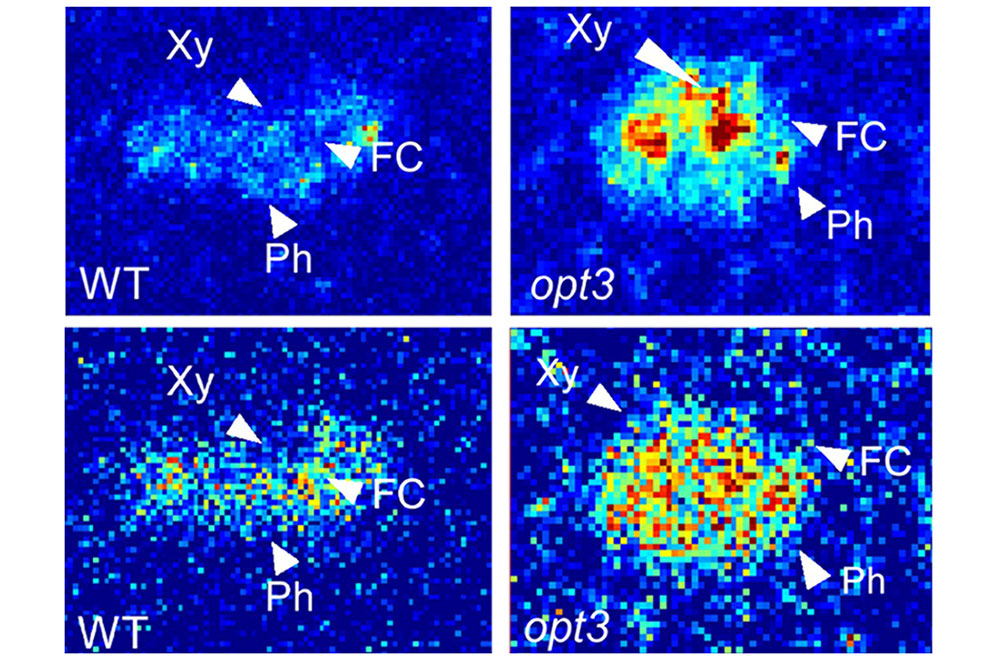
These x-ray "maps" generated at NSLS-II show the distributions of iron (top row) and copper (bottom row) throughout mature leaf petioles, both in unaltered plants (left, WT) and mutant plants (right, opt3). Warm colors (red, orange, and yellow) indicate high concentrations, and cool colors (green and blue) indicate low concentrations. Xy points to the xylem, Ph points to the phloem, and FC points to the fascicular cambium, another type of plant tissue. The maps showed that the mutant plants exhibited larger concentrations of iron in the xylem and, surprisingly, that the copper distribution resembled the iron distribution.
A team of scientists from Cornell University and the U.S. Department of Energy’s (DOE) Brookhaven National Laboratory have revealed an unexpected function of a transport protein and its role in plant regulatory mechanisms. Their research, published in The Plant Cell earlier this year, could help reduce human mineral deficiencies by packing essential micronutrients into edible parts of plants.
Iron is an essential mineral for humans. In addition to being a key component of hemoglobin—the red blood cell protein that carries oxygen throughout the body—iron aids the immune system and plays a role in cognitive functions. The human body cannot produce iron, so it must be consumed regularly. Plants, like spinach, are one source of iron, but their strict regulatory mechanisms prevent minerals from over accumulating because they are toxic to the plant in high concentrations. Scientists, however, have been studying the transport of minerals, like iron, to figure out a way to override these regulatory mechanisms and increase the nutritional value of edible plants.
 enlarge
enlarge
Arabidopsis thaliana plants, like those used in this study, are often used in research because they reproduce quickly and have a short genome that is entirely mapped out. (Joseph Rubino/Brookhaven National Laboratory)
“This story started long ago,” explained Olena Vatamaniuk, a plant biologist from Cornell and head of the lab responsible for this research. Nearly a decade ago, Vatamaniuk and her colleagues published a surprising discovery—a transport protein called oligopeptide transporter 3 (OPT3) is responsible for moving iron within a model plant called Arabidopsis thaliana, rather than the oligopeptides (small peptides) that the transporter was named for.
As part of an earlier study, researchers at the University of Missouri had found that decreasing OPT3 altered the distribution of iron throughout the A. thaliana plant; the roots were showing signs of iron deficiency, despite an abundance of iron in the leaves. This indicated that OPT3’s role was related to the communication of iron status from the leaves, known as the shoot, to the roots.
The two findings were just the start of a complicated story.
“In our latest study, we wanted to use our knowledge of OPT3’s role to figure out how the transporter was related to shoot-to-root signaling,” said Vatamaniuk. Taking a look inside the plants with ultrabright x-rays was the first step—but OPT3 had another surprise in store for the scientists.
Shining Light on Plant Chemistry
When scientists want to figure out what a protein does, they often choose to observe what doesn’t happen when most, if not all, of the protein is removed from a sample. Removing all OPT3 protein would be lethal to the plant species used in this study, so the researchers genetically altered the plants, creating “mutants” with a lower abundance of OPT3 transporters.
Vatamaniuk and her colleagues wanted to look at how the iron distribution throughout the vascular system differed between the mutant and unaltered plants. The researchers were particularly interested in a transport tissue, called the phloem, because they had discovered OPT3 moving iron into this tissue nearly a decade earlier. The phloem typically transports nutrients from areas where they are highly concentrated, known as sources, to areas where they are scarce, known as sinks. This contrasts the xylem vascular tissue, which transports water and nutrients from the roots to the shoot.
One way to analyze iron distributions in tissues and cells is with confocal x-ray fluorescence imaging (C-XRF), a technique recently developed by Cornell beamline scientist Arthur Woll. Like conventional x-ray fluorescence (XRF) imaging, this technique uses bright x-ray light to reveal the locations of different chemical elements within a sample. But the addition of a very tiny, special lens designed by Woll, called a confocal optic, provides depth sensitivity for researchers to quantify the elemental concentrations within specific compartments of thick samples. Researchers at Cornell create these lenses through a process called nanofabrication.
To apply this technique at an ultra-small scale, the Cornell scientists brought their sample to one of the most advanced x-ray light sources in the world, the National Synchrotron Light Source II (NSLS-II). NSLS-II is a DOE Office of Science User Facility at Brookhaven Lab that produces light beams 10 billion times brighter than the sun.
“NSLS-II was the only facility with a bright enough beamline to get us the resolution that we wanted,” explained Ju-Chen Chia, a researcher in Vatamaniuk’s lab and lead author of this paper. “At the time, no other facility could get us the single-micron resolution C-XRF images that we needed.”
 enlarge
enlarge
At the NSLS-II Submicron Resolution X-ray Spectroscopy (SRX) beamline, shown above, scientists focused a ultrabright light beam down to a single square micron. This light illuminated a section of a petiole so the scientists could visualize the distribution of chemical elements throughout the plant vasculature. (Roger Stoutenburgh/Brookhaven National Laboratory)
The research team’s first stop at NSLS-II was the Submicron Resolution X-ray Spectroscopy (SRX) beamline, led by Andrew Kiss. Woll and Kiss situated a series of mirrors to focus the x-ray beam down to a single square micron on a section of a petiole—the part of the plant connecting the leaves to the stem.
The interactions between the x-ray beam and the leaf petiole emitted fluorescent x-ray signals, which propagated through a nanofabricated confocal optic located only one millimeter away before they were recorded by a silicon drift detector.
“This was really challenging from a technical perspective,” noted Kiss. In addition to working with a small beam spot size, the researchers also had to ensure x-rays from only the surface of the leaf petiole were collected. X-rays collected from the depth of the sample would reduce the resolution and effectively blur the image.
The x-ray fluorescence contains characteristic energies that are like fingerprints for each element in the sample. Kiss and the Cornell scientists decoded these x-rays to figure out which elements were in the sample, the concentrations of those elements, and precisely where they were located.
“In the original paper, we proposed that OPT3 is important for loading iron into the phloem,” explained Chia. “So, we thought that if we analyzed the mutant plant vascular tissues using C-XRF, we should see more iron in the xylem but less iron in the phloem of the mutant.”
The researchers found exactly what they were looking for—but their subsequent analyses took them by surprise.
An Unexpected Finding
Some transport proteins move more than one molecule; in plants, iron is often transported with zinc or manganese. So, analyzing the distributions of multiple minerals, in addition to the mineral of interest, is a fairly common practice when conducting x-ray fluorescence experiments.
“Sometimes changing the concentration of one mineral causes a bunch of other concentration changes in plants,” explained Chia. “Iron, copper, zinc, and manganese are all essential minerals for plant growth, so we like to look at all of them at the same time.”
Though it is essential, copper does not typically share transporters with other minerals in plants. That’s why the researchers were especially shocked when they observed changes in the mutant plant’s copper distribution that mimicked those of the mutant’s iron distribution—indicating that OPT3 also transported copper into the phloem.
“If we hadn’t brought our samples to NSLS-II, we never would have considered one transporter moving both iron and copper in a plant,” said Vatamaniuk, emphasizing how unexpected these results were. “It is quite unusual.”
“This work was a great technical accomplishment for the SRX beamline,” noted Kiss. “But it was an even greater demonstration of the expertise and collaboration here at NSLS-II.” Throughout these experiments, Kiss and Woll worked with Ryan Tappero, leader of the X-ray Fluorescence Microscopy (XFM) beamline, where Chia and her colleagues conducted complementary experiments to confirm their findings.
X-ray Vision
At the XFM beamline, the Cornell scientists wanted to visualize the internal distribution of elements throughout the vasculature of embryonic plants, which were contained within mature seeds. Though cutting open the seeds and scanning their surface—like how the scientists studied the leaf petiole with C-XRF—was tempting, cutting the seeds open could cause element redistribution. Exposing the delicate structures to oxygen could also lead to chemical reactions that change their elemental makeup.
 enlarge
enlarge
Researchers used fluorescence signals recorded at the NSLS-II X-ray Fluorescence Microscopy (XFM) beamline, shown above, to derive cross-sectional images of plant seeds without cutting the seeds open. (Brookhaven National Laboratory)
“Just like medical doctors take CT scans of your body without cutting you open, we used x-rays at the XFM beamline to take a ‘chemical’ CT scan of the mineral elements inside the seeds without cutting them open,” Tappero explained.
Medical CT scans rely on a rotating x-ray source and detector to take a series of exposures, from which computers can reconstruct cross-sectional images of internal structures. NSLS-II scientists do not rotate the x-ray beam, so instead they programmed instrumentation to rotate the seed samples in the x-ray beam while recording the x-ray fluorescence signals.
“The seeds were only half a millimeter in diameter, which made them ideal to scan intact,” Tappero explained. As each egg-shaped seed was zapped with ultrabright x-rays, fluorescence signals could radiate out from the center of the seeds to be measured by a silicon drift detector.
After the first exposure, instrumentation rotated the sample by less than one degree so it could be zapped again from another angle. The instrumentation automatically repeated this process until the sample was rotated a full 360 degrees. This technique is called x-ray fluorescence computed microtomography (F-CMT).
F-CMT cross-sectional images are derived from fluorescence signals like conventional XRF images; however, scientists use additional computer reconstruction techniques to provide the cross-sectional views. Using these cross-sectional images to visualize the internal distribution of elements in the embryonic plants, the scientists observed lower concentrations of both iron and copper in the vascular cells of the mutant seeds compared to the unaltered seeds. These results served as further evidence of the OPT3 transporter moving both iron and copper.
“We brought our samples to NSLS-II so we could observe the physiology of this transport protein and we got to come back to our lab with an important piece of the puzzle that lies at the center of it all,” noted Chia. “Everything was about to come together.”
Another Chapter in the OPT3 Story
The researchers returned to their Cornell labs to make sense of their new findings with a deep dive into the mutant plant’s genetics. Through a series of experiments, they discovered that iron and copper not only share a transport protein, but they also interact in a complex signaling pathway that regulates their uptake through gene expression.
This research is just one step towards mitigating human mineral deficiencies by changing the nutrient content of edible plants. Vatamaniuk and her colleagues studied A. thaliana, a non-grass plant that is often used in research because it reproduces quickly and has a short genome that is entirely mapped out. The researchers can now use their findings to look at the function of this transport protein in grass plants like rice, wheat, or barley.
“The physiology of a plant can tweak the function of a transporter,” explained Vatamaniuk. “So, it is important to apply this knowledge to other plants. I’m sure there are more discoveries to come.”
“I want to express gratitude to the NSLS-II scientists because they really help us,” she added. “The nature of collaboration is so important, but they are also just so friendly and helpful.”
“We have so many ambitious ideas,” Chia said, “and they help us bring them to life.”
This study was supported by the National Science Foundation and the DOE Office of Science. Operations at NSLS-II are supported by the DOE Office of Science.
Brookhaven National Laboratory is supported by the Office of Science of the U.S. Department of Energy. The Office of Science is the single largest supporter of basic research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time. For more information, visit science.energy.gov.
Follow @BrookhavenLab on social media. Find us on Instagram, LinkedIn, Twitter, and Facebook.
2023-21431 | INT/EXT | Newsroom









