Q&A with CFN User Xiaowei Teng
To design nanostructured materials for energy conversion and storage applications, Teng of the University of New Hampshire extensively uses the electron microscopy facilities at Brookhaven Lab's Center for Functional Nanomaterials (CFN)
December 20, 2017

Xiaowei Teng
Safe, environmentally friendly, and sustainable—these characteristics are the guiding principles of Xiaowei Teng’s research to design new types of nanostructured materials for energy conversion and storage applications. To design these materials, Teng—an associate professor of chemical engineering in the College of Engineering and Physical Sciences at the University of New Hampshire and principal investigator of the Teng Research Group—needs to characterize their atomic structure and chemical composition in great detail. The powerful beams of x-rays, neutrons, and electrons that are available at the U.S. Department of Energy’s (DOE) national laboratories are uniquely suited for such nanoscale characterizations. As one of five DOE Nanoscale Science Research Centers, the Center for Functional Nanomaterials (CFN)—a DOE Office of Science User Facility at Brookhaven National Laboratory—has played an especially important role in Teng’s research.
Energy storage is a broad area. What does your research in this area focus on?
This research, funded by a DOE Early Career Research Program Award, focuses on electrochemical energy storage using earth-abundant electrode materials in water-based (aqueous) electrolyte solutions, with sodium ions as the carriers of electrical charge moving from one electrode to the other. The biggest advantages of aqueous battery systems compared to conventional non-aqueous ones like lithium-ion batteries are that they are safer and lower cost. Lithium batteries contain very flammable components—the lithium metal and the electrolyte (a solution of lithium ions and organic chemicals) that can combust easily upon contacting water or oxygen in the air. You may recall what happened two years ago with certain brands of phones catching fire. Because of these concerns, a moisture- and air-free environment is required to manufacture the batteries, adding to the cost.
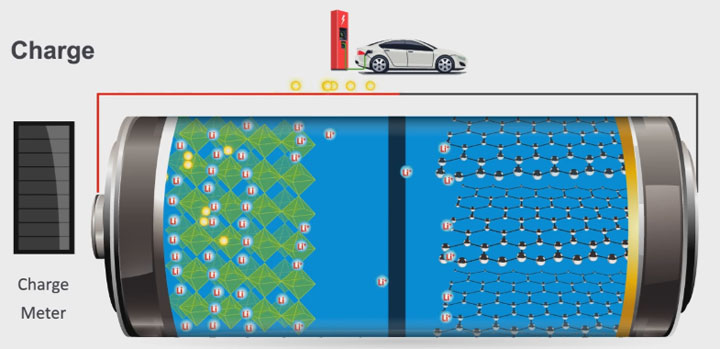
When a lithium-ion battery is charging, lithium ions flow from the positive electrode to the negative electrode through an electrolyte. During discharging, the ions move back to the positive electrode. In both charging and discharging, electrons flow around the external circuit. This movement of electrical charge creates an electrical current through the circuit. Credit: DOE's Office of Energy Efficiency and Renewable Energy; created by Sarah Harman and Charles Joyner. See animation
Beyond the safety and cost issues, lithium resources are not sustainable for the long run. Not many countries have enough abundance to support a future transportation system comprised of electric vehicles. Even optimistic estimates cite that we would have enough lithium to sustain electric vehicle production for only about 50 years.
On the other hand, sodium is widely abundant (about 1,000 times more abundant than lithium in the earth’s crust) and inexpensive (for example, table salt is approximately 40 percent sodium by weight). Additionally, sodium does not have the same manufacturing concerns. However, the storage capacity of aqueous sodium-ion batteries is not as high as lithium counterparts. That is mainly because water-based electrolytes have a narrow voltage window of 1.23 volts. Beyond this voltage, water will decompose into oxygen and hydrogen gases, impairing the battery’s electrode and posing a fire hazard. In our research, we are trying to design new types of materials that are not only highly resistant to water decomposition but also very active for the reduction and oxidation (redox) reactions that are behind the charging and discharging of the battery.
Have you succeeded at designing any materials that are both redox active and highly resistant to water decomposition?
In collaboration with several DOE labs, including Brookhaven, my group recently published a paper in Nature Communications in which we achieved a stable voltage window of over 2.5 volts in a three-electrode (working, reference, and counter electrodes) half-cell using a unique electrode nanomaterial (Mn5O8) in an aqueous solution containing sodium ions. We also achieved a stable voltage window of 3.0 volts in a two-electrode (anode and cathode) full-cell.
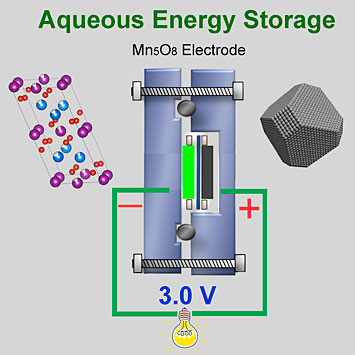
This two-electrode full battery cell—containing both a reduction half-cell and oxidation half-cell—achieved an electrochemically stable voltage window of 3.0 volts. Typically, cells with water-based electrolytes are limited by water's narrow voltage window of 1.23 volts. For aqueous energy storage devices to be competitive with non-aqueous ones, this window needs to be expanded.
This manganese oxide material is not new. A British scientist discovered its structure in 1965. But in the past 50 years, only about 20 papers involving this material have been published, and very few explore the material’s potential for energy storage. At first, we did not know we had this material; we knew it had a unique property but thought it was something else entirely. We asked CFN physicist Dong Su—who has expertise in electron microscopy and works with many people in the battery community in addition to conducting his own battery research—to take a look at our sample with a transmission electron microscope. When he took the image, he found a very strange, atypical structure. At the time, he did not know what it was either. He told us that we might want to look more carefully at our material. So, we took our sample to the Spallation Neutron Source (SNS), a DOE Office of Science User Facility at Oak Ridge National Lab. About three or four months later, we went back to Dong and told him it was a monoclinic type of structure (crystal system with three axes of unequal length, only two of which intersect at right angles with each other) reported almost 50 years ago.
So we got this very exciting electrochemical result about two years ago but spent the last year and a half trying to figure out what the structure was. It is quite unique. It is the only manganese oxide structure that is layered and whose manganese is bivalent, or divalent (Mn ions have valences of +2 and +4, meaning they lose two and four electrons, respectively). Because of its unique bivalent structure (Mn2+2Mn4+3O8), the material forms a solid-liquid “interphase” region via the strong interaction between Mn2+ ions on the surface and water.

Mn5O8 has a unique lattice structure, as seen in (c), with purple denoting Mn4+, blue Mn2+, red O, and white the probability of an unoccupied site. The transmission electron microscopy (d) and scanning transmission electron microscopy (e) images of the Mn5O8 nanoparticles were taken at CFN.
Water bonds so strongly to the surface of this material that it is not energetically favorable for water to decompose to hydrogen and oxygen gases. This strong bonding is the reason why our system operates over the kinetically stable 3.0-volts potential window. We are very excited by our result, as it opens up the possibility of making the performance of aqueous energy storage devices competitive with that of lithium-ion batteries, which usually run under a potential window higher than 3.5 volts.
You also perform research related to energy conversion. What is the goal of that work?
My research group is trying to design electrocatalysts for converting bioethanol into electrical energy. This fundamental work is funded by the National Science Foundation and could have a strong transformative impact on society.
Most people are probably familiar with ethanol as the 10 percent component in their gasoline. This small amount of ethanol oxygenates the fuel and reduces air pollution.
A few years ago, I stopped at a gas station while I was visiting Brookhaven Lab and noticed that it offered E85 fuel, which contains 85 percent ethanol and 15 percent gasoline. I later learned that many gas stations across the country offer E85, which is the fuel that flexible fuel vehicles—such as certain models of the Dodge Dart and Ford Focus—use. Ethanol is a useful chemical in this sense—you pour it into the internal combustion engine and you get heat and power as a result of the combustion of ethanol.
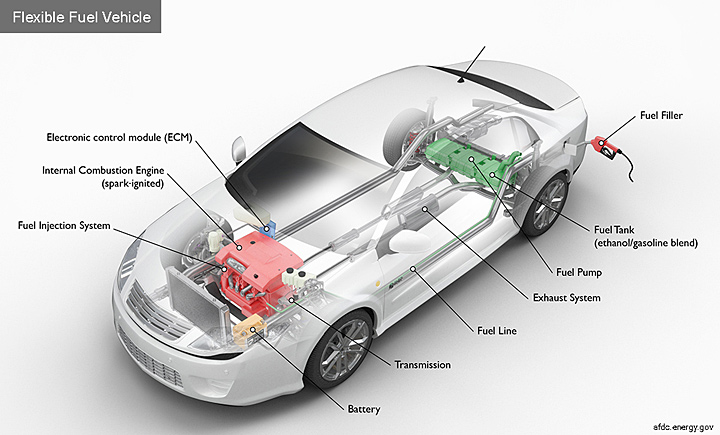
Flexible fuel vehicles have one fueling system, and most components are the same as those found in a typical gasoline car. Some special ethanol-compatible components are required to compensate for the different chemical properties and energy content in ethanol, such as modifications to the fuel pump and fuel injection system. The engine control module is also calibrated to accommodate the higher oxygen content of ethanol. Credit: DOE's Alternative Fuels Data Center.
However, I began thinking about how ethanol could do something even better. As a chemical engineer, I know that electrochemical energy conversion devices (fuel cells, for instance) can obtain thermodynamic efficiencies as high as 95 percent upon the electro-oxidation of ethanol (analogous to ethanol combustion in electrochemical devices). But if you put ethanol in a car and burn it via a thermochemical reaction (typical combustion reaction between ethanol and oxygen gas), it is much less efficient—at most 30 percent (three times less than that of the fuel cell).
Another advantage of ethanol fuel cells compared to hydrogen fuel cells relates to their safety. Because ethanol has a much higher boiling point than hydrogen, it can be more safely stored and transported. Moreover, hydrogen is highly flammable. In the lab, scientists can be careful. But in the real world, accidents are much more likely to happen.
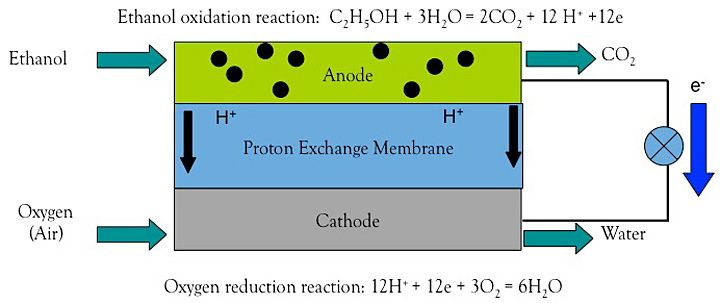
The ethanol fuel cell relies on the oxidation of ethanol at the anode (negative electrode) to produce carbon dioxide, protons (H+), and electrons (e). Protons are transported across a proton exchange membrane to the cathode (positive electrode), where they react with oxygen in the air to produce water. Because ethanol oxidation is slow, Teng and his team are developing catalysts to accelerate the reaction.
The really exciting thing about ethanol is that you can use non-food resources like grasses, wood chips, and other cellulose-based materials to produce it. In addition, crops grown for bioethanol production absorb carbon dioxide emitted into the atmosphere, in sharp contrast to the use of fossil fuels for harvesting energy. Therefore, energy production from bioethanol is considered a sustainable approach. Theoretically, ethanol fuels could replace all petroleum. North America is currently the number-one producer of bioethanol, having produced nearly 15 billion gallons in 2015 alone. Together, the United States and Brazil produce 85 percent of the world's ethanol.
But the problem is that the ethanol electro-oxidation reaction in which ethanol reacts with water to produce carbon dioxide and protons is very slow. For nearly 10 years, my group has been trying to find a catalyst that can accelerate this reaction so electricity can be efficiently generated.
How has the hunt for the perfect catalyst for ethanol oxidation been going so far?

This journal cover shows a transmission electron microscopy image of the catalyst Teng and his collaborators designed with a partially oxidized platinum-ruthenium core, or "heart," and tin oxide shell. The cycle diagram shows the sustainability of bioethanol-based fuel cells: ethanol can be produced through fermentation processes using sugar or cellulose-containing crops, and these crops in turn absorb the carbon dioxide emitted into the atmosphere.
In September 2016, we reported in the journal ChemCatChem a catalyst that converts ethanol into carbon dioxide with high efficiency. This catalyst contains platinum and ruthenium in its core, surrounded by a tin oxide “shell.” Through our collaboration with Stony Brook University, Brookhaven, and other national labs, we found that its structure is a little more complicated than we originally thought—the core is actually mixed with oxygen atoms, so it is partially oxidized. We determined this chemical composition by x-ray absorption analyses at Brookhaven Lab’s former National Synchrotron Light Source (NSLS)—a DOE Office of Science User Facility—and electron energy-loss spectroscopy at CFN.
We are excited about this result because it is the first report of a partially oxidized noble metal that functions as an even better catalyst than the typical metallic noble-metal catalysts. Our catalyst demonstrated a 2.5-fold increase in the rate of carbon dioxide generation toward the ethanol oxidation reaction.
Right now, we are collaborating with the U.S. Army Research Laboratory. The U.S. Army is interested in ethanol but has a very different take than the commercial sector. Often soldiers need to go to places, such as remote islands, that have no resources or infrastructure like electrical grids. Setting up solar panels for energy generation would be too expensive. What they do have are extra boxes of sugar-containing food waste. If fermented overnight, this food waste could produce up to 10 percent ethanol. Then nitrogen gas could be used to expel ethanol vapor out of a fermentation tank for use in fuel cells.
Ethanol is so easy to make via the fermentation process. I come from China, and studies have confirmed that, dating back to at least 4000 years ago, the Chinese were making alcoholic drinks using rice, honey, and grapes. There is also evidence that people in Mesopotamia and Ancient Egypt used fermentation. Even nowadays people make rice wine by simply letting a mixture of rice, yeast, and water sit for a few days.
Ethanol fuel cells could help alleviate the widespread issue of disposing of food waste. You do not need to start making ethanol from scratch by specifically harvesting crops like corn. So much food is thrown away, but what if it was instead recycled to generate ethanol for fuel cells? That would be a way to make the energy conversion process more sustainable.
You mentioned you perform electron energy-loss spectroscopy at CFN. What other electron microscopy techniques do you use at CFN, and how else do you characterize your materials?
We extensively use the electron microscopy facilities at CFN to understand the structure of our materials. Some of the techniques we use are environmental x-ray photoelectron spectroscopy and Raman spectroscopy. Both are techniques for studying how the physical and chemical properties of materials change. The environmental electron microscope allows us to study the materials in realistic scenarios—for example, how our aqueous energy storage materials behave in contact with electrolytes. CFN scientist Xiao Tong of the Interface Science and Catalysis Group has been very helpful.
We combine this structural and chemical information with that collected through x-ray scattering and absorption experiments. We were one of the very active users of Brookhaven Lab’s former NSLS, publishing many papers in collaboration with NSLS and CFN. Now we are using the new NSLS-II beamlines.
We also have performed experiments at the Advanced Photon Source (APS), another DOE Office of Science User Facility at Argonne National Lab, especially when NSLS was shut down and NSLS-II was still under construction. As I previously mentioned, the SNS at Oak Ridge is another facility we use. Because neutrons are much heavier than x-rays and electrons, they can penetrate into materials much more deeply. Neutrons also have a special interaction with lightweight elements like oxygen, hydrogen, and sodium that allows us to have better understanding of a material’s structure when it interacts with water.
Energy materials are complicated, especially at the nanoscale, because there are lots of structural defects and dislocations that impact the performance of the materials. As a result, materials characterization is a challenge during the charge-transfer processes. The electron microscopy facilities at CFN, x-ray beamlines at NSLS-II and APS, and neutron beams at SNS have provided us a great opportunity to characterize the true structure of our materials. Through tailoring structure-property relationships, we can ultimately tailor the structure our materials to achieve the desired performance for energy applications.
How did you come to learn about CFN?
I attended the University of Rochester for graduate school in chemical engineering. One of my senior advisors in the Department of Physics and Astronomy would tell me about how he traveled to Brookhaven all the time for his condensed matter physics work, and he suggested that I check it out.
I ended up doing my postdoc work at CFN just after it opened, and I was one of the first postdocs to work there. The CFN was still being constructed, so I was actually in Brookhaven’s Chemistry Department before we started moving over to the new building in 2007.
So you have been with CFN since the beginning. How have you seen it evolve over the past decade, and what keeps you coming back?
The user program really has been built up over the years. For me, it is the combination of state-of-the-art equipment and trained scientists who know how to operate the equipment so well that makes CFN so valuable to my research. Facilities like those at CFN are not available in university settings because they are too expensive. But just because you have a million-dollar setup does not necessarily mean you have a user-friendly one. The CFN is very unique in this sense. The scientists at CFN understand what you are trying to accomplish through your experiments, and you can have fruitful intellectual discussions with them. CFN scientists collaborate with you, providing useful feedback to improve your experimental design and helping you obtain an in-depth understanding of your data. Communicating with CFN scientists often sparks new ideas.
The CFN has been very accommodating to my schedule, allowing me to pick the preferred date of my visits, as I need to give lectures at my home institution regularly throughout the academic year. I go to CFN at least three or four times a year, staying two or three days at a time. I try to schedule exams while I am gone so that I only have to miss one day of class that I can make up later.

Teng with students in his research group at the University of New Hampshire.
The environment at CFN is especially important for my students. Of course, I want them to learn how to operate the equipment but I also want them to interact with established scientists so they can mature as scientists themselves. In addition to my students performing research at CFN, I bring them every May to the NSLS-II & CFN Joint Users’ Meeting, where I have them present their posters and approach NSLS and CFN scientists to hear about new research. Since I started at UNH in 2009, my group has coauthored 14 papers with Brookhaven scientists.
What is your proudest accomplishment to date?
I would have to say achieving a stable voltage window of over 2.5 volts for aqueous energy storage. This accomplishment really made aqueous energy storage devices competitive with lithium-ion batteries.
Though it is fundamental research, it is very transformative at the same time. A breakthrough is desperately needed to enable the large-scale implementation of renewable energy. Places like Texas and California have so much sunlight that they generate more electricity around noontime than they need. The extra electricity has to be sold to the neighboring states (at a negative price). Without the proper electric energy storage, the amount of electricity generated will not match peak consumption (usually at night). This mismatch puts much stress on the electrical grid, making it unstable. We need a solution for storing all of the energy generated during the day (peak generation) for use at night (peak consumption).
Beyond the electric grid, there are so many applications, such as mobile electronic devices and electrical vehicles. For example, according to Tesla, the cost of a lithium-ion battery in a Tesla Model S is $12K, not to mention what happens after the battery’s lifetime—what should we do with the used battery? It is a known fact that recycling lithium from spent batteries is more expensive than producing it from mining. There is currently no established recycling program for lithium-ion batteries. The old lead-acid batteries in today’s cars can be brought to places like AutoZone for recycling, and you actually receive a merchandise credit when you do so.
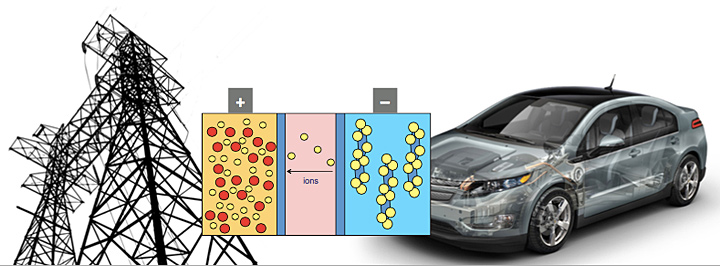
Teng's materials research is part of DOE's effort to develop next-generation batteries and energy storage technology for the electric grid and electric vehicles. Credit: Joint Center for Energy Storage Research, a DOE Energy Innovation Hub.
As I mentioned, lithium-ion batteries are very expensive and unsafe. The aqueous energy storage breakthrough we reported in Nature Communications could offer a safer, more environmentally friendly, and much more resource-abundant option for such applications. In my opinion, nothing can compete with safety—not even price.
Has science always been an interest of yours?
My dad is an engineer. He worked on diesel engines through the navy in China. When I was a kid, he often brought work home and sometimes would bring engine blueprints. Back then, I imagined building things like engines when I grew up, and I actually wanted to become a mechanical engineer when I was a child. But in high school, I liked chemistry lab so much. Seeing how chemistry can turn things different colors and compositions was like magic for me. I thought and still believe now that chemistry really is beautiful. It was then that I decided I would do chemical engineering instead (though I still like the engine very much!).
Brookhaven National Laboratory is supported by the Office of Science of the U.S. Department of Energy. The Office of Science is the single largest supporter of basic research in the physical sciences in the United States, and is working to address some of the most pressing challenges of our time. For more information, please visit science.energy.gov.
Follow @BrookhavenLab on Twitter or find us on Facebook.
2017-12678 | INT/EXT | Newsroom









