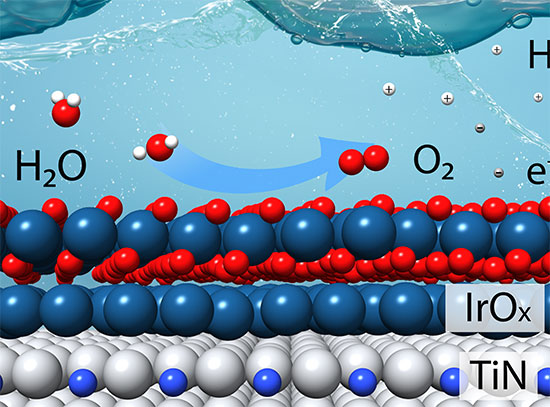
Electrochemical water splitting using electrolyzers is an alternative approach for green hydrogen production, which is the ultimate non-carbon system if uses carbon-neutral electrical energy such as that derived from solar, wind, hydro or nuclear power sources. The current practical membrane electrolyzers utilize acidic electrolytes, and thus there is a need for stable precious metals. The high costs and scarcity of these materials make it difficult to meet the DOE targets for cost-effective hydrogen production.
To address this challenge, the NCWE team seeks to reduce the cost of catalysts in acidic environment using nitrides of Earth-abundant metals (EAM). The state-of-the-art synthesis, characterization theoretical modeling and machine learning methods are strongly coordinated, going from fundamental understandings of catalytic behaviors under operating conditions toward design principles for developing EAM-based materials as active, stable and cost-effective electrocatalysts for water electrolysis.