Narcolepsy Drug Being Used to Improve Cognitive Performance Affects Brain Dopamine Activity
Suggesting Potential For Abuse and Dependence
March 17, 2009
The following news release about the addictive potential of Modafinil, a drug used to treat narcolepsy and increase cognitive performance, was issued by the Journal of the American Medical Association. Note that the content of this news release is embargoed until Tuesday, March 17, 2009, 3 p.m. Central Time (4 p.m. Eastern Time). No story about this research may be printed, posted, or otherwise published prior to that time. A large part of this research was conducted using positron emission tomography (PET) scans at the U.S. Department of Energy’s Brookhaven National Laboratory. Led by Brookhaven senior chemist Joanna Fowler and National Institute on Drug Abuse Director Nora Volkow, the researchers found that Modafinil, a drug used to treat narcolepsy that is used increasingly off label by healthy individuals with the expectation that it will improve cognition, acts similarly to cocaine and methylphenidate – a well-established drug used to treat attention-deficit/hyperactivity disorder. For more information, contact Karen McNulty Walsh at 631-344-8350 or kmcnulty@bnl.gov.
EMBARGOED FOR RELEASE: 3 P.M. (CST) TUESDAY, MARCH 17, 2009Media Advisory: To contact Nora D. Volkow, M.D., call Stephanie Older at 301-594-6145 or email olders@nida.nih.gov.
CHICAGO — Preliminary research in healthy men suggests that the narcolepsy drug modafinil, increasingly being used to enhance cognitive abilities, affects the activity of dopamine in the brain in a way that may create the potential for abuse and dependence, according to a study in the March 18 issue of JAMA.
 enlarge
enlarge
Brookhaven senior chemist Joanna Fowler: "Like cocaine and methylphenidate, Modafinil blocks dopamine transporters thereby increasing dopamine levels in the brain. This study raises awareness about Modafinil’s potential for abuse and addiction in vulnerable populations and signals the need to monitor its use."
Modafinil, a wake-promoting drug used in the treatment of sleep disorders, may enhance cognition and is used off-label for the treatment of cognitive dysfunction in some psychiatric disorders (i.e., schizophrenia, attention-deficit/hyperactivity disorder [ADHD]). The Physicians’ Desk Reference cautions that it can produce psychoactive and euphoric effects typical of central nervous system stimulant drugs, and there is debate surrounding its potential for abuse, according to background information in the article. The mechanisms of action of modafinil are not well understood but are believed to differ from those of stimulant medications (such as methylphenidate and amphetamine), which increase dopamine (a neurotransmitter in the brain essential for the normal functioning of the central nervous system) in the brain by targeting the dopamine transporters, a mechanism that underlies the abuse potential of these drugs. However, there is growing evidence that dopamine may also play a role in the mode of action of modafinil.
Nora D. Volkow, M.D., of the National Institute on Alcohol Abuse and Alcoholism, Bethesda, Md., and colleagues at Brookhaven National Laboratory conducted a study to test whether modafinil, at therapeutic doses, would elevate extracellular (located or occurring outside of cells) dopamine in the brain by blocking the dopamine transporter. The study included 10 healthy men, between the ages of 23-46 years, who received either placebo or modafinil: 200 mg, the dose recommended for narcolepsy; or 400 mg, a dose shown to be beneficial for the treatment of ADHD. The effects of modafinil on extracellular dopamine and on dopamine transporters were measured by positron emission tomography (a radiographic technique used to examine biochemical activity in tissue).
The researchers found: “In this pilot study, modafinil acutely increased dopamine levels and blocked dopamine transporters in the human brain. Because drugs that increase dopamine have the potential for abuse, and considering the increasing use of modafinil for multiple purposes, these results suggest that risk for addiction in vulnerable persons merits heightened awareness.”
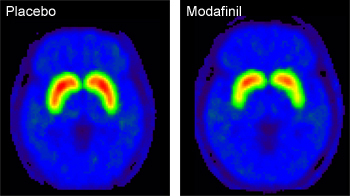 enlarge
enlarge
PET images of the brain show that subjects given modafinil had lower levels of the radiotracer [11C]raclopride bound to dopamine receptors than subjects given a placebo. Red represents the highest amount of binding. This signal indicates higher levels of dopamine release in the modafinil subjects. (Dopamine competes with the radiotracer so higher levels of dopamine "push" the tracers out.)
Modafinil also increased dopamine in the nucleus accumbens, a brain region critical for the rewarding effects of drugs of abuse.
“Modafinil was developed with an expectation that a medication could have a non-dopaminergic target for its wake-promoting effects. However, the current findings in humans, along with preclinical studies, documenting the indispensable role of dopamine in the wake-promoting effects of modafinil, support modafinil’s dopamine-enhancing effects as a mechanism for its therapeutic actions.”
(JAMA. 2009;301[11]:1148-1154. Available pre-embargo to the media at http://www.jamamedia.org)
Editor’s note: This research was supported by the DOE Office of Biological and Environmental Research, National Institutes of Health, National Institute on Alcohol Abuse and Alcoholism, National Institute of Biomedical Imaging and Bioengineering, and the General Clinical Research Centers. Please see the article for additional information, including other authors, author contributions and affiliations, financial disclosures, funding and support, etc.
2009-10926 | INT/EXT | Newsroom









