Understanding the Initiation of Protein Synthesis in Mammals
March 18, 2014
 enlarge
enlarge
Messenger RNA (in red) latches closed around a pre-initiation complex, and attaches to transfer RNA (in green), beginning a process of protein synthesis specific to eukaryotes — animals, plants, and fungi.
Protein synthesis, the process by which cells generate new proteins, is the most important cellular function, requiring more than 70 percent of the total energy of a cell. The initiation of this process is the most regulated and most critical component, but it is still the least understood.
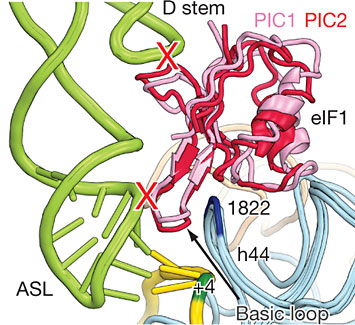
Research by Ivan Lomakin and Thomas Steitz of Yale University has unlocked the genetic scanning mechanism that begins this crucial piece of cell machinery.
They determined the structures of three complexes of the ribosome, a complex molecular machine that links together amino acids to form proteins according to an order specified by messenger RNA. These three structures represent distinct steps in protein translation in mammals – the recruitment and scanning of mRNA, the selection of initiator tRNA, and the joining of large and small ribosomal subunits.
“Any small defect or disruption in the protein synthesis process can cause abnormalities or disease,” said Lomakin. “Understanding this process is critical for understanding how human life comes to be, and how some over-expressions or abnormalities in the initiation of protein synthesis may be connected to cancer or Alzheimer’s or other diseases.”
"Understanding this process is critical for understanding how human life comes to be."
— Ivan Lomakin
Using x-ray crystallography on ribosomal subunits purified from rabbit cells, they were able to determine the positions and roles of the different pieces of cellular machinery, a bit like creating a playbook for a football game. They found that the tRNA and mRNA compete for position at the P site – one of three key sites on a ribosome – where short chains of amino acids are linked to form proteins.
“Now, we have a low resolution structure, so we can’t yet talk about atomic details of the mechanism,” said Lomakin. “The next important step is to get higher resolution images. And we can change organisms to see if they behave differently, so we’re working on the structure of human ribosome initiation complexes, too.”
For this higher resolution, Lomakin and his collaborators will use the National Synchrotron Light Source II, a new state-of-the-art light source that will begin early science at Brookhaven National Laboratory in 2014. “Our hope is to be able to look at very weak diffraction to get higher resolution structures of these important cellular mechanisms.”
This work was supported by the National Institutes of Health (NIH) grant GM022778 (to T.A.S.)
2014-4718 | INT/EXT | Newsroom









