10 Questions with Gayani Perera
interview with a CFN Scientist
April 21, 2014

Gayani Perera
At its most extreme, nanoscience involves synthesizing and characterizing materials at the atomic-scale, exploring fundamental properties and engineering entirely new technologies. This billionth-of-a-meter tailoring often demands a precise portrait of the way atomic surfaces interact and transform during chemical reactions.
Gayani Perera, a Postdoctoral Research Associate at the proximal probes facility of Brookhaven Lab’s Center for Functional Nanomaterials, specializes in these real-time investigations. Perera uses a technique called scanning tunneling microscopy (STM) to reveal sub-nanometer details and help understand and enhance nanomaterials.
How does scanning tunneling microscopy work?
In 1981, scientists developed STM to push visualizations way beyond the limits of traditional optical microscopes. In STM, an extremely sharp needle called a tip travels over a material’s surface. When we bring that tip to within one nanometer of the surface and apply a small bias voltage, electrons cross that tiny gap—this phenomenon is known as "tunneling.”
As the tip scans laterally across the sample surface, an electrical feedback mechanism controls its vertical position over the sample surface to maintain constant tunneling current. Under the right conditions, we can measure the surface textures down to individual atoms. With precise STM control and data acquisition, we can translate the tunneling data into images.
What distinguishes your STM techniques and expertise?
Surface science usually explores materials in ultra-high vacuum (UHV) and cryogenic temperatures, conditions far from those in catalytic chemistry. But the morphology, structure, chemical composition, and reactivity of a catalyst under those controlled conditions can differ drastically from its native operating environment. Probing catalysts under realistic operating conditions is necessary and, in fact, very exciting.
Using CFN’s Reactor STM we can see surface reactions with pressures and temperatures that are typically present in real-world applications. With in situ examinations of surfaces under catalytic conditions in real time we can really unravel the process on a basic level. This unique capability helps us discover novel effects that will be essential to understanding surface chemistry and heterogeneous catalysis.
How do you apply that real-time imaging to catalysts?
The Reactor STM allows us to prepare and characterize single-crystal surfaces under UHV conditions, after which they are transferred into a catalytic flow reactor to image surface structure and to follow the catalyzed transformation of reactants into products simultaneously.
For example, macrocyclic molecules with single metal centers are promising candidates for heterogeneous catalysts with well-defined active sites. Alkene epoxidation using manganese–porphyrin, which we examine at CFN, is one system ideally suited for real-time STM.
Using reactor STM experiments, it’s now possible to relate catalyst structure with catalytic activity, either by means of gas analysis or by imaging the reaction turnover at the active site. With the reaction tracked, scientists can improve the catalyst design.
You push STM techniques beyond pure imaging—what does that involve?
Other than obtaining atomic resolution images, the STM tip can be used to accurately manipulate single atoms or molecules, by pushing or dragging them at low temperatures. This fascinating technique can be used to fabricate atomic-scale structures or to perform basic chemical reaction steps, such as dissociation and bond-formation processes. By combining STM manipulations with the tunneling spectroscopy, the chemical, electronic, and mechanical properties of single molecules can be studied at each reaction step.
Why did you come to Brookhaven Lab and CFN to do this research?
I love doing research, and this laboratory is a perfect fit for my interests. Moreover, cutting-edge experimental tools, techniques, and expertise can all be found under one roof, along with a great group of dedicated colleagues. The proximity to other facilities, staff scientists, and users makes it very easy to collaborate and interact across disciplines in ways that you might not typically imagine for your research.
Describe one recent experiment or collaboration that you found particularly interesting.
As I mentioned before, I’m currently working on the interaction of small gas molecules such as H2 and O2 with macrocyclic compounds containing single metal centers, whose reactivity may be tuned via different bonding environments. Such studies have mostly been limited to UHV and cryogenic conditions that are far from applied catalytic chemistry.
We performed initial Reactor STM experiments to probe manganese-porphyrin on gold, mimicking the function of the natural enzyme-cytochrome P450. We use simultaneous imaging and gas analysis in Reactor STM to investigate activity and selectivity. Ultimately, we envision expanding these experiments to macrocyclic species with other metal centers.
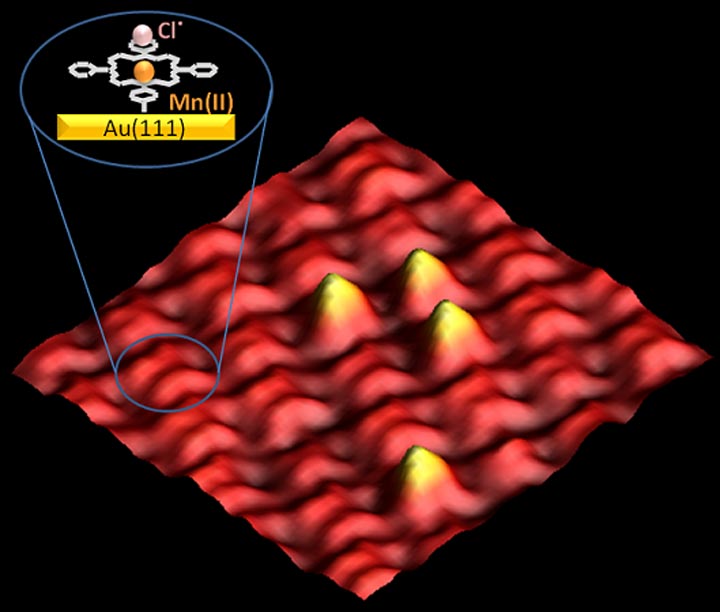
STM image of a monolayer of manganese-porphyrin self-assembled on a gold surface after exposure to 0.8 bar O2 at room temperature. STM measurements revealed only sporadic O2 dissociation, suggesting inefficient dissociation of the Cl• radical at 300K. Bright spots correspond to the interaction with oxygen.
What are some of the biggest experimental challenges involved?
STMs can be difficult to use effectively, and there are a few very specific techniques that require a lot of skill and precision. STMs require very stable and clean surfaces, excellent vibration control, and sharp tips. The quality of that tip is crucial for improvement of the spatial resolution and the reliable interpretation of experimental data. Sometimes these tips can break or become unstable, which results in flawed STM images.
What personally excites you about your research?
The thrill of discovery! There's always the possibility of seeing something novel that no one else has seen before. This excitement drives me to work even harder, especially with always-advancing technology and a facility with cutting-edge resources.
Do you have any advice for incoming users or scientists interested in taking advantage of CFN staff and facilities?
At CFN you can be exposed to all the expertise you need. As a postdoctoral associate, CFN provides a great place to improve my skills within a short period of time. I’ve also been very lucky to benefit from state-of-the-art facilities that are not just a collection of different high-end instruments, but also a place where scientists and professional experts are available and ready to discuss new challenges and provide advice and solutions. There are always opportunities to collaborate—inter-disciplinary collaboration really is the culture of the laboratory.
Why did you decide to become a scientist?
I was always good at math and science, so I just followed that path. I fell in love with physics in high school and was also fortunate to have excellent teachers. I earned my undergraduate degree in physics with a minor in mathematics from the University of Colombo, Sri Lanka, with highest distinction. Then I decided to pursue that field all the way through my Ph.D. in Condensed Matter Physics at Ohio University in 2011 before I joined the interface and catalyst group at CFN.
2014-4820 | INT/EXT | Newsroom









