- Home
-
Research Groups
Division Groups
- Artificial Photosynthesis
- Catalysis: Reactivity & Structure
- Electrochemical Energy Storage
- Electron- and Photo-Induced Processes for Molecular Energy Conversion
- Neutrino and Nuclear Chemistry
- Surface Electrochemistry and Electrocatalysis
Associated Groups
- Catalysis for Alternative Fuels Production
- Nanostructured Interfaces for Catalysis
- Structure and Dynamics of Applied Nanomaterials
- People
- Operations
- News
- Events
Artificial Photosynthesis
Mechanisms for CO Production from CO2 Using Re(bpy)(CO)3X Catalysts
The chemical conversion of CO2 has been studied by numerous experimental groups. Particularly, the use of rhenium tricarbonyl based molecular catalysts has attracted interest owing to their ability to absorb light, store redox equivalents, and convert CO2 into higher-energy products. The mechanism by which these catalysts mediate reduction, particularly to CO and HCOO–, is poorly understood, and studies aimed at elucidating the reaction pathway have been hindered by the large number of species present in solution. We investigated the mechanism for carbon monoxide production using rhenium tricarbonyl catalysts using density functional theory (DFT). The investigation presented is based on the experimental work of Meyer’s group as well as our group. Their work with a simplified reaction mixture, one that removes the photo-reduction step, is used for validation of the proposed mechanism, which involves formation of a rhenium carboxylate dimer, [Re(dmb)(CO)3]2(OCO).
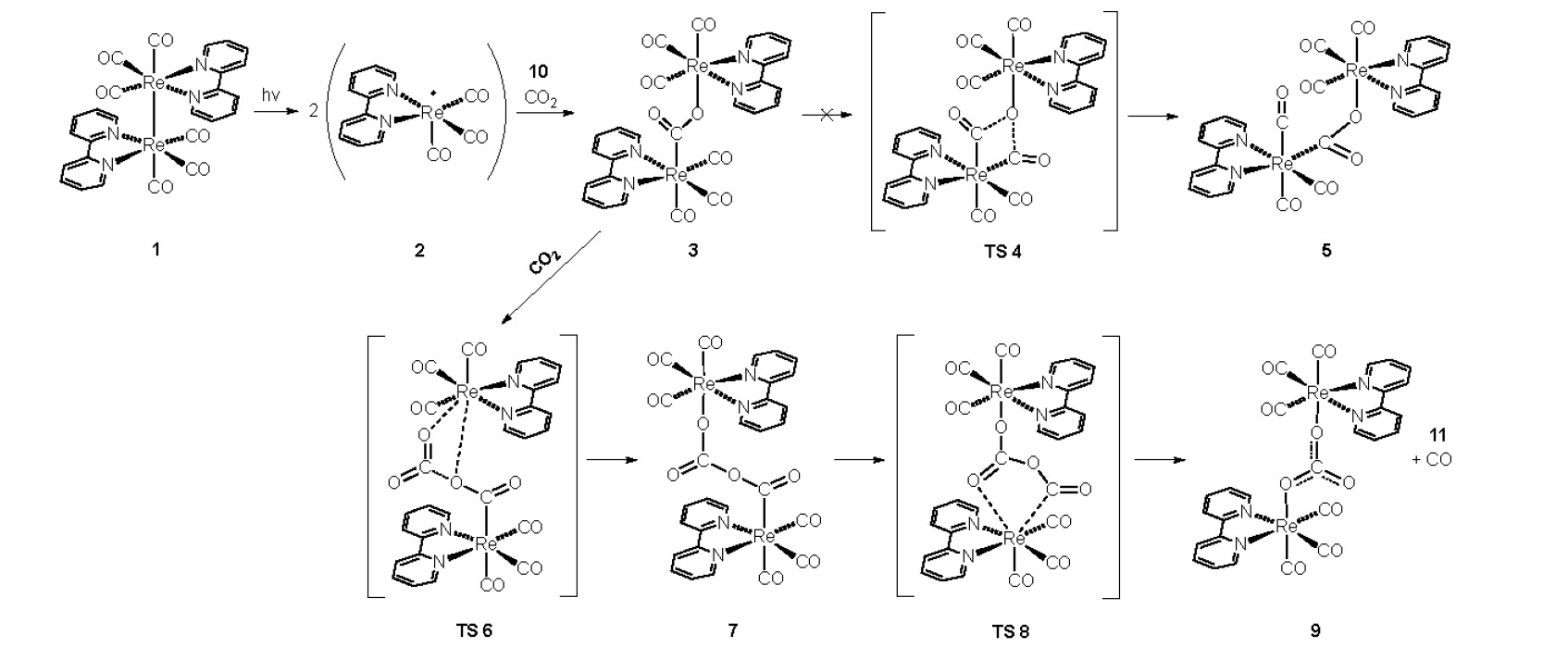
CO2 insertion into this species, and subsequent rearrangement, is proposed to yield CO and the carbonate bridged [Re(dmb)(CO)3]2(OCO2). Structures and energies for the proposed reaction path are presented and compared to experimental observations.
J. Am. Chem. Soc. 2012, 134, 5180-5186, DOI: 10.1021/ja2105834.




