- Home
-
Research Groups
Division Groups
- Artificial Photosynthesis
- Catalysis: Reactivity & Structure
- Electrochemical Energy Storage
- Electron- and Photo-Induced Processes for Molecular Energy Conversion
- Neutrino and Nuclear Chemistry
- Surface Electrochemistry and Electrocatalysis
Associated Groups
- Catalysis for Alternative Fuels Production
- Nanostructured Interfaces for Catalysis
- Structure and Dynamics of Applied Nanomaterials
- People
- Operations
- News
- Events
Nanostructured Interfaces for Catalysis
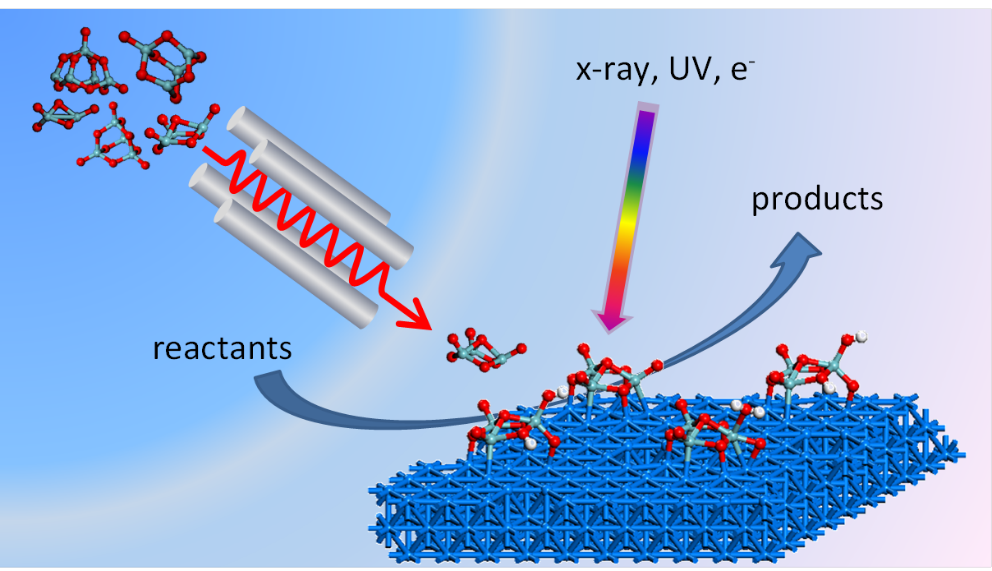
The Nanostructured Interfaces for Catalysis group is part of the BNL catalysis science program, which advances fundamental understanding of sustainable fuel and chemical synthesis pathways with a current emphasis on catalysis for C1 chemistry such as conversion of CO2. The goal of the Nanostructured Interfaces for Catalysis research is to identify and characterize the catalytically active sites of nanostructured surfaces and explore how these are influenced or can be manipulated by variations in composition, particle size, morphology and chemical environment (e.g., support, reaction conditions).
These goals are pursued by theory and experiment on model systems, both planar and powder, whose surface structure/morphology, and chemical composition are well-defined. We are particularly interested in exploiting the unique electronic and chemical properties of ultra-small clusters (1-50 atoms) of metals and metal compounds (oxides, sulfides) as novel catalytic materials. Cluster-based nanocatalysts are prepared by mass-selected deposition of gas-phase clusters which provides control over size and chemical composition with atomic precision.
The model nanocatalysts are tested for conversion reactions involving CO2 and methane, with an emphasis on revealing the atomic structure at interfaces and the interfacial electronic properties that underlie observed catalytic activity. In this way, our research program provides atomic-level descriptions of catalyst structure and activity that informs in situ and operando studies of complex catalysts used in reactor systems.
Group Leader
-
Michael White
Professor of Chemistry, State University of New York at Stony Brook, Senior Chemist
(631) 344-4345, mgwhite@bnl.gov





