Does Stimulant Treatment for ADHD Increase Risk of Drug Abuse?
Researchers say treatment age and duration can influence outcome; urge further study
June 18, 2007
UPTON, NY — Parents, doctors, and others have wondered whether common treatments for attention-deficit hyperactivity disorder (ADHD) inadvertently predispose adolescents to future drug abuse. The answer may depend on the age at which treatment is started and how long it lasts, say the authors of a new brain-imaging and behavioral study conducted in animals at the U.S. Department of Energy's Brookhaven National Laboratory. The results appear in the June 5, 2007 online issue of the journal Pharmacology, Biochemistry and Behavior.

"Our study shows that the brain's reward pathways are definitely influenced by methylphenidate, one of the stimulant drugs commonly used to treat ADHD," said Brookhaven researcher Panayotis (Peter) Thanos, lead author of the study. "But the brain chemistry changes we observed suggest that the developmental stage at which treatment begins and the duration of treatment are important variables that need further study."
In the study, rats were given methylphenidate mixed with distilled water beginning one month after birth — early adolescence for rats. Animals received either 1 or 2 milligrams methylphenidate per kilogram of body weight, consistent with clinical doses given to children with ADHD. A control group of rats was handled under identical conditions but given plain water.
After two months of treatment, and again after eight months, the scientists performed positron emission tomography (PET) scans to measure the levels of dopamine D2 receptors, a type of brain receptor important for experiencing reward and pleasure that has been linked to pleasure and drug abuse. After the eight-month treatment, animals were also tested for their propensity to self-administer cocaine.
Rats given the 2mg/kg dose of methylphenidate were significantly less likely to press a lever to self-administer cocaine, and received fewer self-initiated infusions of the drug following eight months of treatment than the lower-dose group or the control rats.
The changes observed in brain chemistry were specific to the age and duration of methylphenidate treatment: Specifically, after two months of treatment, brain scans revealed that both groups of treated rats had lower levels of dopamine D2 receptors in their brains than did control animals.
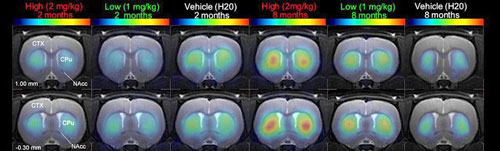 enlarge
enlarge
MicroPET brain images of rats show that two months of daily 2 mg/kg methylphenidate treatment caused a decrease in dopamine D2 receptor availability in the brains of treated rats compared with control rats, while 8 months of treatment had an opposite effect, increasing dopamine D2 receptor availability. (Click image to enlarge.)
In contrast, after eight months of treatment, the brain scans revealed elevated levels of dopamine D2 receptors in treated rats compared with controls, with the higher-dose treatment group showing the highest level of D2 receptors. In the control group, D2 receptor levels declined with age. Research at Brookhaven and elsewhere has suggested that low levels of dopamine D2 receptors may increase the likelihood of drug abuse, while elevated levels of dopamine D2 receptors may attenuate the propensity to abuse drugs.
"This new study provides evidence that chronic methylphenidate treatment begun in adolescence affects the brain's dopamine D2 receptor levels, and thus the brain's reward circuitry, differently depending on the age and treatment duration," Thanos said. The scientists' observation of lower rates of cocaine self-administration in the animals treated for eight months with a 2kg/mg dose of methylphenidate supports this idea.
However, the observation of lower levels of D2 receptors after two months of treatment suggests that shorter lengths of treatment or the age at which treatment is evaluated could result in different effects. "Lower dopamine D2 receptor levels following short-term treatment could make the animals more vulnerable to drug self-administration during early adulthood," Thanos said. "Unfortunately, we cannot compare cocaine self-administration following eight months of treatment with that obtained after two months of treatment in the same animals, since animals were not tested for cocaine self-administration at this earlier time," Thanos said. "We wanted to avoid any confounding effect that might have resulted from cocaine exposure during this early developmental stage," he explained.
Evaluating the effect of treatment duration is one avenue the researchers are exploring in follow-up studies "to help assess optimal duration of treatment regimes to minimize adverse effects on the propensity to abuse drugs," Thanos said.
Thanos notes that the findings from this study cannot be directly extrapolated to treatment regimes used for ADHD. Also, these studies were done in healthy animals, not in rodent models of ADHD. All experiments were conducted in conformity with the National Academy of Sciences Guide for Care and Use of Laboratory Animals and Brookhaven National Laboratory Institutional Animal Care and Use Committee protocols.
This research was funded by the National Institute on Alcohol Abuse and Alcoholism intramural program and by the Office of Environmental and Biological Research within the U.S. Department of Energy's Office of Science.
2007-10659 | INT/EXT | Newsroom









