Added Robust Interphase Stabilizes Promising Cathode Material
Scientists demonstrated a 97% charge retainment after 200 cycles, opening the pathway to longer-lasting batteries
August 10, 2022
 enlarge
enlarge
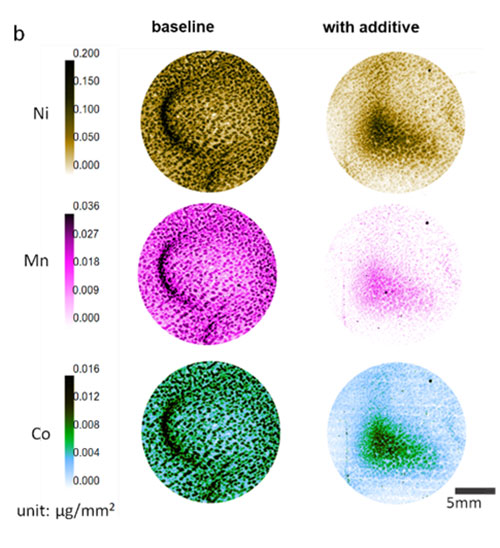
The image shows the different elements that can be found in the interphase and how they are distributed. (Image courtesy of Nat Energy 7 (6), 484-494 (2022).)
The Science
Scientists demonstrated that adding lithium difluorophosphate (LiDFP) to nickel-rich layered cathode materials (NMC) mitigates cathode degeneration effectively during high-voltage cycling, retaining a charge of 97% after 200 cycles (baseline of 77.3%).
The Impact
NMCs are promising next-generation battery materials; however, their practical capacities are far less than the theoretical values due to their structural instability during cycling, especially when charged at high voltages.
Summary
From cell phones to cars, lithium batteries are an integral part of everyday life and our demands on stored energy continue to rise. To meet this demand, researchers are working on next-generation batteries. Nickel-rich layered materials (lithium nickel manganese cobalt oxides, also known as NMCs) are a promising candidate due to their high theoretical capacity, high working voltage, and relatively low cost. However, serious capacity fading occurs for NMCs when subjected to high-voltage charging, which should be addressed to take full advantage of NMCs. In this study, a team of researchers collaborated with scientists at the National Synchrotron Light Source II (NSLS-II) to find a solution for this.
By adding lithium difluorophosphate to a common commercial electrolyte, the team demonstrated the formation of a robust interphase layer that effectively mitigated the strain and crack formation responsible for the degradation process. In their experiment the team showed that a cathode using this additive would retain 97% of its original charge after 200 cycles with high-voltage cycling. By using a multimodal research approach at the NSLS-II involving four beamlines, as well as beamlines at the European Synchrotron Radiation Facility, they showed that the protective layer was formed by additive decomposition and how it also suppressed transition metal dissolution as well as cathode surface reconstruction. The four beamlines they used were In situ and Operando Soft X-ray Spectroscopy (IOS), Quick X-ray Absorption and Scattering (QAS), Submicron Resolution X-ray Spectroscopy (SRX), and X-ray Powder Diffraction (XPD). All four beamlines are part of the advanced materials characterization suite available at NSLS-II. As a U.S. Department of Energy Office of Science User Facility, NSLS-II offers access to its experimental stations through a free peer-reviewed access process.
This research opens the pathway for stabilizing NMCs or other layer-structured materials via electrolyte engineering, an approach that may pave the way for next-generation energy technologies.
Download the reserach summary slide (PDF)
Contact
Yijin Liu
SLAC National Accelerator Laboratory
yijinliu@stanford.edu
Xiao-Qing Yang
Brookhaven National Laboratory
xyang@bnl.gov
Kang Xu
US Army Research Laboratory
conrad.k.xu.civ@army.mil
Enyuan Hu
Brookhaven National Laboratory
enhu@bnl.gov
Publications
Sha Tan, Zulipiya Shadike, Jizhou Li, Xuelong Wang, Yang Yang, Ruoqian Lin, Arthur Cresce, Jiangtao Hu, Adrian Hunt, Iradwikanari Waluyo, Lu Ma, Federico Monaco, Peter Cloetens, Jie Xiao, Yijin Liu, Xiao-Qing Yang, Kang Xu, Enyuan Hu. Additive engineering for robust interphases to stabilize high-Ni layered structures at ultra-high voltage of 4.8 V. Nature Energy 7 (6), 484-494 (2022). DOI: 10.1038/s41560-022-01020-x
Funding
S.T., Z.S., X.-Q.Y. and E.H. are supported by the Assistant Secretary for Energy Efficiency and Renewable Energy, Vehicle Technology Office of the US Department of Energy (DOE) through the Advanced Battery Materials Research Program, including the Battery500 Consortium under contract no. DE-SC0012704. S.T., Z.S., X.-Q.Y. and E.H. also acknowledge the support from the Assistant Secretary for Energy Efficiency and Renewable Energy, Vehicle Technology Office of the US DOE through the Applied Battery Research for Transportation programme under contract no. DE-SC0012704. HAADF-STEM measurements and DFT computational work used the resources of the Center for Functional Nanomaterials, a US DOE Office of Science User Facility at Brookhaven National Laboratory, under contract no. DE-SC0012704. This research used beamlines 5-ID, 7-BM, 23-ID-2 and 28-ID-2 of the NSLS II, a US DOE Office of Science user facility operated for the DOE Office of Science by Brookhaven National Laboratory under contract no. DE-SC0012704. The hard X-ray phase contrast tomography was conducted at the nano-imaging beamline ID16A-NI at the European Synchrotron Radiation Facility, Grenoble, France. A.C. and K.X. thank the Joint Center of Energy Storage Research, an energy hub funded by the US DOE Office of Science, for support. Y.L. and J.L. acknowledge the support from the US DOE Laboratory Directed Research and Development program at SLAC National Accelerator Laboratory under contract DE-AC02-76SF00515.
2022-20882 | INT/EXT | Newsroom