Decoding the Structure of Petroleum with Yunlong Zhang
interview with a CFN user
September 22, 2020
 enlarge
enlarge
ExxonMobil chemist Yunlong Zhang holds a structural model of petroporphyrin, a molecule found in crude oil. Since 2018, Zhang and Percy Zahl—a scientist at Brookhaven Lab's Center for Functional Nanomaterials—have been using high-resolution atomic force microscopy (HR-AFM) to study petroleum chemistry. By analyzing HR-AFM images, they developed a "guide" for discriminating pollutant-causing sulfur and nitrogen atoms in well-known molecules of petroleum. Their research was featured on the cover and published in an Energy & Fuels issue in 2019.
Yunlong Zhang is a staff scientist focusing on hydrocarbon science and petroleum chemistry in Corporate Strategic Research at ExxonMobil Research and Engineering Company. Starting in 2018, his research to decipher the structure of petroleum molecules brought him to the Center for Functional Nanomaterials (CFN) at the U.S. Department of Energy’s (DOE) Brookhaven National Laboratory. Here, in the CFN Proximal Probes Facility, exists a specialized microscope for directly visualizing individual molecules. Zhang received a PhD in physical organic chemistry from The Ohio State University in 2010, after obtaining his master’s and bachelor’s degrees in organic and medicinal chemistry in China. His research has led to more than 70 publications—including peer-reviewed articles, reviews, and book chapters—as well as patents, invited talks, and awards.
ExxonMobil is one of one of the largest publicly traded energy companies in the world. Perhaps lesser known is ExxonMobil’s long history of corporate research. How does research and development (R&D) fit into the company’s history and mission?
ExxonMobil has been engaged in R&D since the company started more than a century ago. The Corporate Strategic Research (CSR) lab in Clinton, New Jersey, where I work, was established to conduct long-term, transformative energy research. Two years ago, we celebrated the 50th anniversary of CSR and the numerous scientific discoveries and breakthroughs that unfolded here. A case in point is the work of M. Stanley Whittingham—one of the three awardees of the 2019 Nobel Prize in Chemistry—who pioneered the development of lithium-ion batteries here in the 1970s and early 1980s.
In addition to the Clinton site and labs in Baytown and Houston, Texas, ExxonMobil has research labs in Europe, Asia, and Canada. Research conducted at these centers has helped provide the energy that is fundamental to our daily lives—from the clean, efficient fuels for transportation to the natural gas for power generation and heating homes and businesses.
In the two decades since the merger of Exxon and Mobil, the company has strengthened its commitment to fundamental research. In addition to its internal research programs, ExxonMobil has invested in strategic collaborations through major energy centers at MIT, Princeton, UT Austin, Stanford, and academic institutions abroad. Recently, the company started a broad agreement with DOE national labs that allows ExxonMobil scientists to work with top experts and access research capabilities at these facilities.
ExxonMobil’s current research focuses on developing next-generation renewable energy and sustainability solutions, including advanced biofuels, carbon capture and storage technologies, natural gas technologies, and new energy-efficiency processes.
How did you come to join ExxonMobil?
I have always been very interested in applying fundamental scientific research to solve real-world problems. After obtaining my PhD degree in chemistry, I joined Robert Langer’s lab at MIT as a postdoctoral fellow researching biomaterials for drug delivery. There, we engineered new ways to package large biological molecules—such as proteins and DNA—for nanomedicine. My research focused on designing biocompatible materials that are responsive to internal or external stimuli. A few of the materials we designed were patented and licensed to biotech companies for further development.
Bob really inspired me to embrace challenges and come up with innovative solutions with maximum impact. After my postdoc, I joined CSR at ExxonMobil because it seemed to provide an ideal environment for doing both fundamental and applied research on practical and technical problems, with the potential to impact our economy and our world.
How did you make the transition from designing drug delivery systems to studying petroleum?
It seems like quite a big change in research fields, but my experiences at MIT significantly broadened my skills and knowledge. Most importantly, I learned how to collaborate with people from diverse backgrounds. These interactions enabled me to learn the “languages” spoken by different fields to solve problems, transition to a new field, and conduct interdisciplinary research.
Now, as part of the Hydrocarbon Science Section of the CSR lab at ExxonMobil, I work side by side with many chemical and mechanical engineers. As a physical organic chemist, I bring unique perspectives. Of course, I had a lot to learn in the petroleum field. But my training has prepared me to contribute new ideas to some very old problems—like figuring out the structure of petroleum molecules.
Why do we need to know the structure of petroleum molecules?
Many of the fuels and nonfuel products that we depend on in our daily lives—from gasoline, diesel, jet fuel, and natural gas to lubricants, asphalt, fertilizers, detergents, plastics, polymers, and even medicines—are produced from petroleum. Petroleum is nonrenewable; it is known colloquially as a “fossil fuel,” as it was formed hundreds of millions of years ago from the remains of living organisms under geological conditions.
The crude oils coming out of the ground contain many impurities. Besides petroleum, they contain water, clays and minerals, and chemicals and surfactants, which are added to aid in oil extraction. Crude oils first need to be refined to remove impurities and then distilled into different fractions based on their boiling points. These fractions are largely the feedstocks for making gasoline, diesel, and jet fuel, but they need to be refined further before they’re suitable for sale. While petroleum molecules are mostly (more than 96 percent by weight) composed of carbon and hydrogen, they also contain small amounts of other atoms (heteroatoms) such as sulfur, nitrogen, oxygen, and even metals. These heteroatoms can produce emissions when they combine with oxygen during combustion of fuels and generate sulfur and nitrogen oxides, which lead to acid rain and smog. Much of the complex chemistry in a refinery focuses on removing these heteroatoms by catalytic processes.
Knowing the structure of petroleum molecules is of paramount importance to all petroleum industrial processes. Problems in oil production can be caused by waxes, gas hydrates (ice-like solids made of water and gas), or asphaltenes (heavy organic compounds) because of changes in temperature and pressure. Assurance of flow of petroleum requires selection of just the right chemical additives. Transportation pipelines or the distillation tower can be corroded by acidic molecules in petroleum. In addition, some minor elements contained in petroleum—such as transition metals or heteroatoms—can deactivate the catalysts.
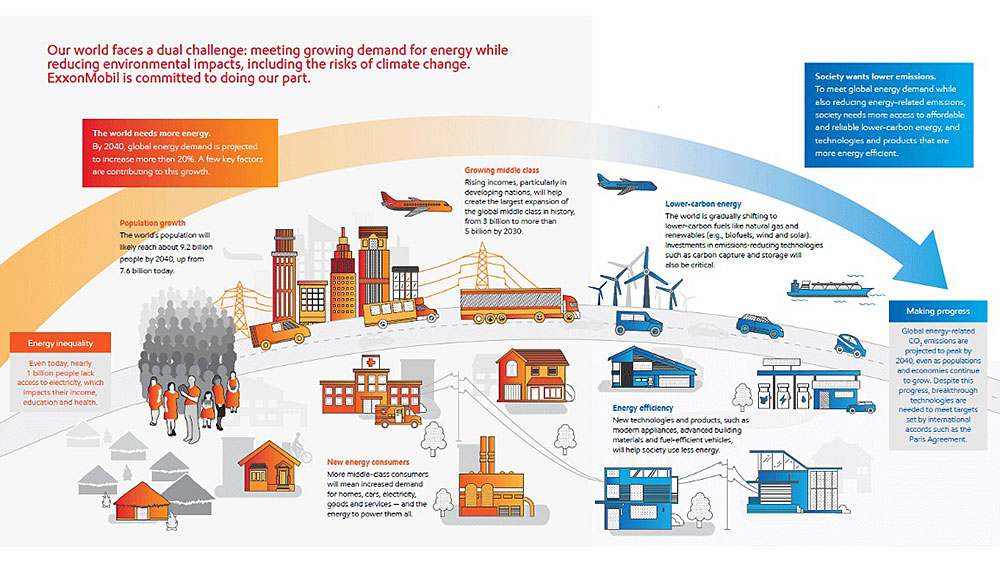
Most importantly, knowing the structure of petroleum molecules will enable us to find new uses of petroleum to help solve the dual challenge we are facing. The premise of the dual challenge is as follows: on one hand, there is a growing demand for energy from an increasing world population with improved living standards; on the other hand, we must simultaneously mitigate the risks of climate change.
Nuclear energy and renewables such as wind, solar, and biomass can all potentially be part of the solution when it comes to alternative sources of power. However, challenges remain when considering our need for feedstock materials that build the products we rely on every day. The world still needs metals such as steel and concrete for infrastructure and lithium for batteries. Currently, the extraction and production of these resources consumes a significant amount of energy and often generates excess carbon dioxide emissions. With greater understanding of the structure of petroleum molecules, perhaps we can harness these compounds to form creative solutions for the materials demand. For example, today, airplanes can mostly be made from composite plastic and carbon fiber materials, which are derived from petroleum sources. Of course, this approach illustrates one of many in addressing this dual challenge. Like a puzzle, the full solution will require a combination of many elements.
 enlarge
enlarge
A schematic showing the dual energy challenge of today. Source: Exxon Mobil, "Innovating Energy Solutions: Research and Development Highlights."
In the United States, petroleum has been used as an energy source since the 19th century. Why do we still not understand its structure?
First, petroleum is one of the most complex substances on the planet because it contains a daunting number of molecular structures. Technological advances over the past century have enabled us to identify an increasing number of molecules, but we still have a long way to go.
Since it was invented in the early 1900s, x-ray crystallography has been a primary tool for characterizing molecular structures. But a small crystal is required for this technique to work. The crystalline structure diffracts the x-ray beam in a way that reveals the 3-D arrangement of atoms in the sample.
In the 1950s, the development of mass spectrometry, nuclear magnetic resonance, and other structural characterization techniques overcame this requirement. However, these techniques are best suited for pure compounds—those containing identical molecules. When that’s not the case, scientists often adopt a “divide and conquer” strategy: purify a substance into its components and individually characterize their molecular structures. Because petroleum contains so many compounds, it would be impossible to purify crude oils into pure fractions or crystals.
Nonetheless, many generations of chemists and engineers have studied petroleum to the extent possible with the existing tools. These studies—largely based on the divide-and-conquer strategy—formed the foundation of our knowledge on petroleum today. They would first separate petroleum into several fractions and then characterize each fraction by deducing a representative or average structure (because each fraction is still a complex mixture).
Recently, scientists using the most advanced mass spectrometry technique available detected more than half-a-million peaks in a single droplet of oil. And these peaks only distinguish between molecules with different masses, or molecular weights. Many different molecules could have exactly the same mass. Thus, an astronomical number of possible structures could be associated with one mass. For example, C40H82 has 6.2 x 1013 isomers, or compounds with the same molecular formula but different atomic arrangements.
Thus, the structure of petroleum molecules had only been derived indirectly. Many in the scientific community thought it would be impossible to obtain the structure directly.

The first-ever direct image of a chemical structure—specifically of pentacene, an aromatic hydrocarbon made of five rings fused in a linear arrangement. Credit: IBM.
But that idea changed in 2009, when IBM scientists from Zurich published a paper in Science reporting the first direct picture of an organic (carbon-containing) molecule. For many chemists, including me, it was a dream come true: seeing a physical image of a molecule, just like a drawing. I still like to look at this image; it’s as breathtaking now as it was then.
How were these IBM scientists able to do the “impossible?”
They used a technique called noncontact atomic force microscopy (nc-AFM) to image the structure of the molecule at very high resolution (HR-AFM). For HR-AFM, you don’t need to purify samples. You just take a look. Physicist Richard Feynman presented this idea during a lecture he gave in 1959: “It would be very easy to make an analysis of any complicated chemical substance; all one would have to do would be to look at it and see where the atoms are.” Over the past few decades, scientists have built instruments to do exactly that. In particular, HR-AFM comes in handy to solve petroleum structures, for which there is no way to extract a single molecule in a large enough quantity to characterize it using conventional methods.
How does HR-AFM work?
The capability to image individual molecules by HR-AFM relies on many advances made by solid-state physicists since the invention of AFM in the 1980s. nc-AFM and HR-AFM belong to the family of scanning probe microscopy techniques, which include both AFM and scanning tunneling microscopy. As its name suggests, nc-AFM is operated in noncontact mode, meaning that the microscope probe does not touch the sample. Most AFMs employ contact or tapping modes.
The no-touch operation is enabled by a sensor called a tuning fork, which is made of quartz. Attached to the very end of the cantilever is a single molecule, often carbon monoxide (CO). This CO functionalized tip scans over a single layer of molecules on a flat surface. The molecular interaction forces between the tip and sample cause a frequency shift in the oscillation of the cantilever. By mapping this frequency shift, we can obtain images of the molecules. Using ultrasmall sensing amplitudes of just a few picometers (pm) and a single inert (unreactive) molecule at the probe apex, HR-AFM has a high sensitivity.
An animation showing the noncontact atomic force microscopy (AFM) technique in high-resolution AFM operation mode with a carbon monoxide probe molecule flexing in close proximity to a sample molecule.
HR-AFM is very delicate and challenging. The instrument must be cooled down to about 5 Kelvin—just a few degrees above absolute zero—to ensure the molecule under investigation stands still (avoids thermal drifting and vibrations). This cooling also reduces thermal noise in the detector to a level that allows us to measure tip oscillations that are smaller than an angstrom, which is one hundred-millionth of a centimeter. The instrument also needs to be operated in ultrahigh vacuum (below 10-10 mtorr), without the presence of interfering air molecules. The low temperature and pressure are essential for generating images of molecules, atoms, or even subatomic scales.
How did you discover the HR-AFM capability at the CFN?
In 2017, I attended the 20th International Conference on NC-AFM in Suzhou, China. There, I met CFN staff scientist Percy Zahl, who was the only other U.S. attendee. I learned that Percy did his postdoc in the same IBM group in Zurich and was optimizing hardware and software for HR-AFM. Very few groups in the United States are working on HR-AFM, so I was surprised to hear this. Percy told me that he had upgraded an HR-AFM instrument at the CFN—which is only 120 miles away from the CSR lab where I work—and was interested in testing it with small organic molecules.

Percy Zahl with the HR-AFM instrument at the CFN. This instrument represents one of a few in the United States.
Later that year, I visited the CFN and discussed my petroleum research in more detail with Percy and other members of the CFN Interface Science and Catalysis Group. Back at ExxonMobil, I proposed that I collaborate with Percy, and I received approval to take advantage of this great opportunity. In the process, I discovered that there was a longtime collaboration between Brookhaven and ExxonMobil on neutron scattering at Brookhaven’s former National Synchrotron Light Source.
Percy and I started to work together in 2018. I propose the chemistry problems and molecules to study; he brings in physics knowledge and performs the experiments. The collaboration has been quite productive. We published one paper in 2019 and have since been working on a few more papers.
What have you learned from the HR-AFM studies at the CFN?
I have had a long-standing interest in understanding how heteroatoms are bonded into petroleum molecules and how this bonding affects the reactivity of these molecules. So, our first idea was to use nc-AFM to see what heteroatoms look like. As I mentioned, heteroatoms are a source of pollutants. So, studying the types and locations of heteroatoms in molecules is important to making cleaner fuels.
Initially, we weren’t sure whether HR-AFM could distinguish these heteroatoms, which have small differences in the number of electrons and nuclei. We used HR-AFM to image the chemical structure of a few sulfur- and nitrogen-containing molecules expected to be in petroleum. By analyzing the AFM images we obtained, we were able to generalize some “rules,” or a guide, for identifying sulfur and nitrogen atoms and distinguishing them from carbon atoms.
For example, the relative size of the elements can be used to distinguish different elements. Sulfur is significantly larger than nitrogen and carbon and thus is relatively easy to pinpoint. However, nitrogen is only slightly smaller than carbon, making it very difficult to tell them apart. Fortunately, we can measure forces to confirm a nitrogen atom if one is suspected. The two nitrogen-containing molecules we studied (carbazole and acridine) can be distinguished by their different ring structures (five- versus six-membered rings). These findings on known molecules will help us apply HR-AFM to characterize unknown molecules in petroleum.

The illustration featured on the June 2019 Energy & Fuels cover shows how HR-AFM can distinguish sulfur- and nitrogen-containing molecules commonly found in petroleum. One discriminating feature is the relative size of the elements, indicated by the size of the boxes in the overlaid periodic table.
Coincidentally, 2019—the year we published our findings in Energy & Fuels—was the International Year of the Periodic Table, which celebrated the 150th anniversary of chemist Dmitri Mendeleev discovering a way to organize the elements according to certain “rules.” Our paper was highlighted on the journal cover to celebrate this event. It attracted considerable attention on Twitter and LinkedIn and received many reads shortly after publication.
In a related effort, we have been trying to link structure to reactivity by studying how carbazole, acridine, and dibenzothiophene (a sulfur-containing molecule in petroleum) interact with metal surfaces. Through these studies, we can learn how these molecules interact with catalyst sites during hydroprocessing (adding hydrogen) to remove sulfur, nitrogen, and other contaminants in petroleum, for example.
We’ve also started exploring several polycyclic aromatic hydrocarbons. These well-known organic molecules are the largest component of petroleum molecules. They’re made of multiple fused rings (polycyclic) and are highly stable structures due to strong bonding arrangements (aromatic). By understanding their aromaticity and bonding character, we hope to address some long-standing chemistry problems and gain insights into industrial processes.
As a relatively newer technique, HR-AFM is still advancing. What are some of the current limitations that must be overcome for HR-AFM to achieve its full potential?
First of all, HR-AFM remains a tool in the hands of a small group of dedicated and specialized physicists with a deep understanding of solid-state physics. Though imaging each molecule takes about 30 minutes, preparing and operating the system and samples in ultrahigh vacuum and at ultralow temperatures is time consuming and expensive. For example, we can only image 30 to 50 molecules for each sample—a process that takes about a month. By comparison, mass spectrometry can capture hundreds of thousands of peaks simultaneously.
Ideally, we’d like the HR-AFM instrument to be stable enough to run for longer times with minimal supervision, scan much faster, and automatically collect data. These capabilities are especially important for my research in characterizing petroleum molecules; 30 to 50 images for a sample are too limited for me to link individual molecules to bulk phenomena and ultimately to discover common or rare structural patterns. However, Percy has made impressive progress in advancing HR-AFM capabilities. For example, he can now run the instrument remotely from home once it’s tuned and stable.
Sometimes the molecules are very “slippery” on the surface during scanning, moving around like ice hockey pucks. This movement generates blurry images, akin to taking a photo with a camera while your hands are shaking. When the molecules move, you have to start all over again. Even when you do get a decent image, it’s difficult to interpret 3-D molecules without comparing them to known structures. And we’re still relying on human perception and judgment to interpret the images. A lot of experience, knowledge, and time is needed to understand the images and find the nuances. Complicated effects arise from various interactions between the tip and sample, and sometimes “artifact bonds”—when the images seem to show a bond that doesn’t really exist—appear. That’s why we’re characterizing more model compounds to tell different heteroatoms or ring structures apart. As more and more molecules are studied by research groups around the world, hopefully a machine learning algorithm can be developed to help interpret the images. However, currently there is a limited number of molecules, and thousands or more images are needed to train the model.
Right now, HR-AFM only works for a small range of molecules with the “right” weight, boiling point, volatility, and shapes. For example, if the molecules are too light, they will be lost by the vacuum pumping and move on the surface during scanning. If they’re too heavy, it may be impossible for us to deposit them onto a surface. Currently, the molecular weight needs to be less than one thousand or above two hundred for us to obtain reliable images. Scientists have long suspected that petroleum contains larger molecules with molecular weights above one thousand.
We need to develop additional ways to deposit samples. Today, we mostly rely only on vaporization via flash heating. Molecules that don’t evaporate easily are problematic. When you think of petroleum, you probably think of oil, a liquid. But most of the heavy molecules in petroleum are actually solids—just like the materials used for paving roads or making roofing shingles. To volatilize and deposit the samples, Percy and I plan to explore whether we can borrow some techniques from mass spectrometry, which encountered similar hurdles during its development. Initially, we plan to test the techniques of laser and electric field desorption.
What is most exciting to you about this research?
While Mendeleev is most well-known for designing the Periodic Table, he also investigated the composition of petroleum and played a major role in founding the first petroleum refinery in Russia in 1879. He is quoted with saying that burning petroleum as a fuel “would be akin to firing up a kitchen stove with bank notes.” In other words, Mendeleev recognized that petroleum was a valuable source from which useful chemicals could be made.
It’s very interesting to look at where we are today, after all of these years. We’ve been burning petroleum to get energy, without really caring too much about its structure. Of course, chemists have wanted to know the structure, but there hasn’t been a direct way to do so until HR-AFM. Though HR-AFM is still in the early stages, we’re already seeing its power. It’s opened a whole new field for studying molecules in complex mixtures. Seeing molecular images from HR-AFM is breathtaking, especially when we find something unexpected or new. I look forward to characterizing more molecules in petroleum with HR-AFM and ultimately figuring out how to piece these molecules together to make useful materials that will improve our lives—my dream since my graduate and postdoc training days.
The CFN and NSLS (and its successor, NSLS-II) are DOE Office of Science User Facilities.
Interested in becoming a CFN user? Submit a proposal through the online proposal system. The next deadline is September 30. If you have questions about using CFN facilities or partnering with CFN scientists, please contact CFN Assistant Director for Strategic Partnerships Priscilla Antunez at (631) 344-6186 or pantunez@bnl.gov.
Brookhaven National Laboratory is supported by the U.S. Department of Energy’s Office of Science. The Office of Science is the single largest supporter of basic research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time. For more information, visit https://energy.gov/science.
Follow @BrookhavenLab on Twitter or find us on Facebook.
2020-17450 | INT/EXT | Newsroom









