New Halide Electrolytes for Solid State Batteries
Scientists find new, cost-effective halide solid electrolytes for use in applications such as electric vehicles
September 30, 2021
 enlarge
enlarge
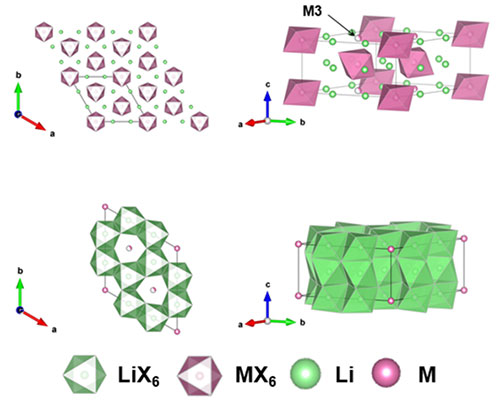
The image shows the atomic crystal structure and Li+ migration pathways that enhance the Li-ion transport through the new halide solid electrolyte material. Image credit: Energy Mater., 11, 2003190 (2021)
The Science
Scientists found new, cost-effective halide solid electrolytes for all-solid-state batteries that are free of rare-earth metals.
The Impact
Halide solid electrolytes (superionic conductors) offer both excellent (electro)chemical stability and the ability to form good solid-solid contacts for solid-state Li batteries. Until now, the use of expensive metals has hindered their practical application.
Summary
To increase the reach of electronic vehicles, researchers are looking for safer batteries with a higher energy density. Solid-state batteries are a potential candidate due to their high energy density and their low risks such as low flammability. One especially interesting type of solid-state battery uses halide-based electrolytes. These halide solid electrolytes offer excellent (electro)chemical stability while maintaining good mechanical sinterability, making them a possible game changer for developing all-solid-state batteries. However, the use of expensive metals hinders their practical applicability.
In this study, a team of researchers reported new halide superionic conductors that are free of rare-earth metals. The two conductors are Li2ZrCl6 and Fe3+-substituted Li2ZrCl6. The benefit of these new conductors is that they use of zirconium (Zr) instead of yttrium (Y) and erbium (Er). Zr is about 10 times more abundant than the other usual candidates.
The research team synthesized the conductors using a mechanochemical method followed by a combination of heat treatments and ball-milling to create a special atomic crystal structure. This specific atomic structure, called hexagonal close-packed, offers a higher conductivity than other halide solid electrolytes.
To investigate the structural and electronic nature of their sample, the scientists used a combination of research tools. Among these tools, the team used the Pair Distribution Function (PDF) beamline at the National Synchrotron Light Source II (NSLS-II). The PDF beamline is part of the advanced research capabilities offered by NSLS-II, a U.S. Department of Energy (DOE) Office of Science user facility located at DOE’s Brookhaven National Laboratory.
Based on the combined research methods, the researchers revealed the evolution of the solid-solution phase in the atomic structure and indicated that the lithium-ion migration is favorable, which in turn is the reason for the high conductivity. They also proved the superior interfacial stability of their material compared to conventional halide solid electrolytes.
Their results demonstrate a breakthrough in practical all-solid-state technologies and provide an intriguing viewpoint for designing new halide superionic conductors.
Download the research summary slide
Contact
Kyung-Wan Nam
Dongguk University
knam@dongguk.edu
Yoon Seok Jung
Yonsei University
yoonsjung@yonsei.ac.kr
Publications
H. Kwak, D. Han, J. Lyoo, J. Park, S. H. Jung, Y. Han, G. Kwon, H. Kim, S.-T. Hong, K.-W. Nam, Y. S. Jung. “New Cost-Effective Halide Solid Electrolytes for All-Solid-State Batteries: Mechanochemically Prepared Fe3+-Substituted Li2ZrCl6,” Advanced Energy Materials, 11, 2003190 (2021) DOI: 10.1002/aenm.202003190
Funding
This research was supported by the Technology Development Program to Solve Climate Changes and by Basic Science Research Program of the National Research Foundation (NRF) funded by the Ministry of Science & ICT (grant no. NRF-2017M1A2A2044501, 2018R1A2B6004996, and 2017M1A2A2044502), by the Technology Innovation Program (grant no. 20007045) funded by the Ministry of Trade, Industry & Energy (MOTIE, Korea). The PDF research used beamline 28-ID-1 (PDF) of the National Synchrotron Light Source II, a US Department of Energy (DOE) Office of Science User Facility operated for the DOE Office of Science by Brookhaven National Laboratory under contract no. DE-SC0012704. The facility support from beamline 16A1 of Taiwan Light Source (TLS) is also acknowledged.
2021-19270 | INT/EXT | Newsroom









