- Home
-
Research Groups
Division Groups
- Artificial Photosynthesis
- Catalysis: Reactivity & Structure
- Electrochemical Energy Storage
- Electron- and Photo-Induced Processes for Molecular Energy Conversion
- Neutrino and Nuclear Chemistry
- Surface Electrochemistry and Electrocatalysis
Associated Groups
- Catalysis for Alternative Fuels Production
- Nanostructured Interfaces for Catalysis
- Structure and Dynamics of Applied Nanomaterials
- People
- Operations
- News
- Events
Electrochemical Energy Storage
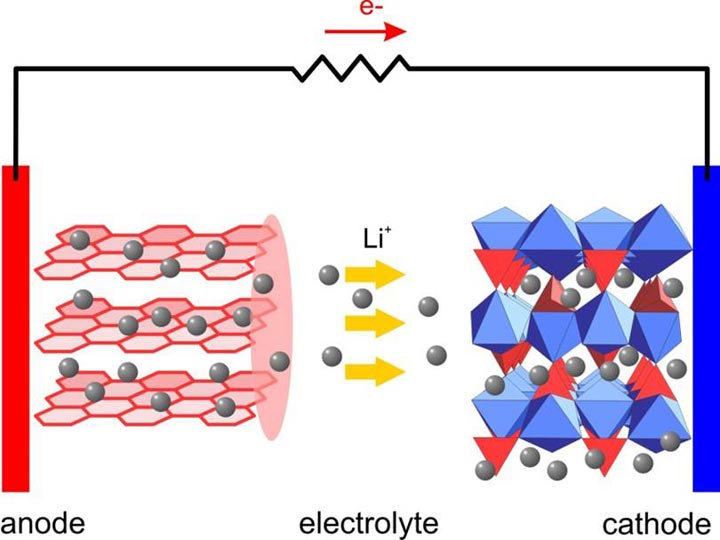
Our research activities are focusing on the development and diagnostic studies of new electrochemical energy storage systems, especially for vehicle applications, and new materials for these systems. In order to meet the challenges of powering different types of electric vehicles in short term, advanced Li-ion batteries (LIBs) with high energy and high power density, low cost, good abuse tolerance, and long calendar and cycle life must be developed. In addition, various beyond lithium-ion new systems such as Li-Air, Li-S, Na-ion batteries need to be explored for long term strategy. Despite current advances, much more R&D will be needed to achieve the performance and lifetime requirements for deployment of these advanced technologies. In order to overcome these barriers, the fundamental understanding of the structural changes of the cathode and anode materials in the bulk and at the surface, the interactions between the electrode material and the electrolyte, the formation and functionality of the SEI on the anode and cathode, the effects of the additives and the surface coatings are critical for advanced Lithium-ion battery systems. Another important research field of this group is the development of advanced diagnostic characterization techniques to investigate these issues, obtaining in-depth fundamental understanding of the mechanisms governing the relationship between the structure and the performance, aimed to provide guidance and approaches to solve these problems. We have been developing and applying the following diagnostic techniques for electrochemical energy storage systems using combined synchrotron based in situ X-ray techniques (x-ray diffraction, hard and soft x-ray absorption) with other imaging and spectroscopic tools such as high resolution transmission electron microscopy (HRTEM), Scanning transmission electron microscopy (STEM), mass spectroscopy (MS), X-Ray fluorescence microscopy (XRF) and transmission x-ray microscopy (TXM).
Advanced Li-ion battery studies
For advanced Li-ion battery materials, we have been studying the new generation of cathode materials such as high voltage spinels (LiMn2-xNixO4) and Li-rich high energy density Li1+x(NiMnCo)O2 composite materials. For the high voltage spinel, the high voltage charge can cause serious electrolyte decomposition and electrode degradation problems. We have been working on the development of new electrolytes using new solvents, new salts, and new additives through organic synthesis, especially the boron based anion receptors. The interaction between the electrolyte and cathode surface, and the effects of cathode surface coating have been studied using synchrotron based XRD and XAS during cycling and through the comparative studies of cells before and after long time cycling. Another important issue relating to battery safety characteristics is the thermal stability of new cathode materials. This problem is being studied using the combined TR-XRD, TR-XAS with mass spectroscopy (MS).
Li-S battery studies
Li-S battery is considered as a very promising system with high energy density and low cost potential. The main problem is the cycling life due to the dissolution of S during cycling through the formation of various lithium polysulfide compounds. We are working on the design and synthesis of polymerized sulfur containing organic compounds to reduce the S dissolution problem during cycling. This is a collaborative research with Prof. Deyang Qu at University of Wisconsin at Milwaukee. Our group has developed a novel in situ X-ray fluorescence (XRF) microscopy combined with X-ray absorption spectroscopy (XAS) technique, which enables us to track the morphology and redistribution of S during cycling.
Sodium-ion battery studies
Sodium-ion batteries (Na-ion) are considered as promising alternatives for Li-ion batteries, especially in large scale applications due to the low cost and abundant nature of Na resources. Unlike the Li-ion batteries, to which electrode materials had been well developed, the suitable cathode and anode materials for Na-ion batteries are quite limited. In collaboration with Prof. Yongsheng Hu and Prof. Hong Li at Institute of Physics, Chinese Academy of Sciences (IOPCAS), we are actively studying the new electrode materials for Na-ion batteries. Most of these materials are synthesized at IOPCAS and their structural changes during charge-discharge cycling are studied at BNL using synchrotron based XAS and XRD.
Li-Air battery studies
Li-air batteries have been identified as a promising battery system. One big problem of Li-air battery is the electrochemical reduction of molecular O2 is known to form superoxide ions (O2-), which can react with organic solvents and specifically carbonate solvents. The decomposition of the solvents induced by O2- consumes the electrolyte as well as orming undesired side-products (e.g. Li2CO3), which are difficult to be re-oxidized during the recharge process. By collaborating with Prof. Deyang Qu at with Prof. Deyang Qu at University of Wisconsin at Milwaukee, the boron-based Lewis acid tris(pentafluorophenyl) borane (TPFPB) was used to solve this problem. It was found that found that TPFPB is a good catalyst for the disproportionation of O2-. The kinetics for the catalytic O2- disproportionation is much faster than the direct chemical reaction between PC and O2-. Therefore, with the addition of TPFPB, the consumption of PC by the O2- produced from the reduction of O2 can be suppressed.




