Tiny Nanocubes Help Scientists Tell Left from Right
New method could improve drug development, optical sensors and more
June 26, 2013
UPTON, NY—In chemical reactions, left and right can make a big difference. A "left-handed" molecule of a particular chemical composition could be an effective drug, while its mirror-image "right-handed" counterpart could be completely inactive. That's because, in biology, "left" and "right" molecular designs are crucial: Living organisms are made only from left-handed amino acids. So telling the two apart is important—but difficult.
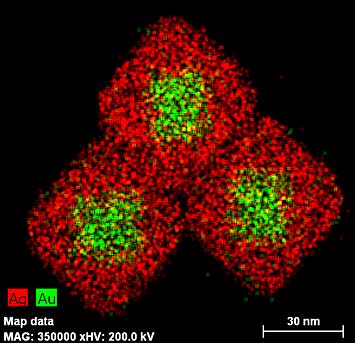
Electron microscopy "maps" of octahedral gold nanoparticles surrounded by cubic silver shells. Attaching a biomolecule (e.g., DNA) to these nanoparticles strengthens a signal representing a difference between left- and right-handed molecules' response to light by 100 times, and pushes it toward the visible range of the electromagnetic spectrum.
Now, a team of scientists at the U.S. Department of Energy's Brookhaven National Laboratory and Ohio University has developed a new, simpler way to discern molecular handedness, known as chirality. They used gold-and-silver cubic nanoparticles to amplify the difference in left- and right-handed molecules' response to a particular kind of light. The study, described in the journal NanoLetters, provides the basis for a new way to probe the effects of handedness in molecular interactions with unprecedented sensitivity.
"Our discovery and methods based on this research could be extremely useful for the characterization of biomolecular interactions with drugs, probing protein folding, and in other applications where stereometric properties are important," said Oleg Gang, a researcher at Brookhaven's Center for Functional Nanomaterials and lead author on the paper. "We could use this same approach to monitor conformational changes in biomolecules under varying environmental conditions, such as temperature—and also to fabricate nano-objects that exhibit a chiral response to light, which could then be used as new kinds of nanoscale sensors."
The scientists knew that left- and right-handed chiral molecules would interact differently with "circularly polarized" light—where the direction of the electrical field rotates around the axis of the beam. This idea is similar to the way polarized sunglasses filter out reflected glare unlike ordinary lenses.
Other scientists have detected this difference, called "circular dichroism," in organic molecules' spectroscopic "fingerprints"—detailed maps of the wavelengths of light absorbed or reflected by the sample. But for most chiral biomolecules and many organic molecules, this "CD" signal is in the ultraviolet range of the electromagnetic spectrum, and the signal is often weak. The tests thus require significant amounts of material at impractically high concentrations.
The team was encouraged they might find a way to enhance the signal by recent experiments showing that coupling certain molecules with metallic nanoparticles could greatly increase their response to light. Theoretical work even suggested that these so-called plasmonic particles—which induce a collective oscillation of the material's conductive electrons, leading to stronger absorption of a particular wavelength—could bump the signal into the visible light portion of the spectroscopic fingerprint, where it would be easier to measure.
Methods based on this research could be extremely useful for the characterization of biomolecular interactions with drugs, probing protein folding, and in other applications where stereometric properties are important.
— Oleg Gang
The group experimented with different shapes and compositions of nanoparticles, and found that cubes with a gold center surrounded by a silver shell are not only able to show a chiral optical signal in the near-visible range, but even more striking, were effective signal amplifiers. For their test biomolecule, they used synthetic strands of DNA—a molecule they were familiar with using as "glue" for sticking nanoparticles together.
When DNA was attached to the silver-coated nanocubes, the signal was approximately 100 times stronger than it was for free DNA in the solution. That is, the cubic nanoparticles allowed the scientists to detect the optical signal from the chiral molecules (making them "visible") at 100 times lower concentrations.
"This is a very large optical amplification relative to what was previously observed," said Fang Lu, the first author on the paper.
The observed amplification of the circular dichroism signal is a consequence of the interaction between the plasmonic particles and the "exciton," or energy absorbing, electrons within the DNA-nanocube complex, the scientists explained.
"This research could serve as a promising platform for ultrasensitive sensing of chiral molecules and their transformations in synthetic, biomedical, and pharmaceutical applications," Lu said.
"In addition," said Gang, "our approach offers a way to fabricate, via self-assembly, discrete plasmonic nano-objects with a chiral optical response from structurally non-chiral nano-components. These chiral plasmonic objects could greatly enhance the design of metamaterials and nano-optics for applications in energy harvesting and optical telecommunications."
This research was conducted at the Center for Functional Nanomaterials and funded by the DOE Office of Science and by the National Science Foundation.
The Center for Functional Nanomaterials is one of the five DOE Nanoscale Science Research Centers, premier national user facilities for interdisciplinary research at the nanoscale supported by the U.S. Department of Energy, Office of Science. Together the NSRCs comprise a suite of complementary facilities that provide researchers with state-of-the-art capabilities to fabricate, process, characterize and model nanoscale materials, and constitute the largest infrastructure investment of the National Nanotechnology Initiative. The NSRCs are located at DOE's Argonne, Brookhaven, Lawrence Berkeley, Oak Ridge, Sandia and Los Alamos National Laboratories. For more information about the DOE NSRCs, please click here.
DOE's Office of Science is the single largest supporter of basic research in the physical sciences in the United States, and is working to address some of the most pressing challenges of our time. For more information, please visit science.energy.gov.
One of ten national laboratories overseen and primarily funded by the Office of Science of the U.S. Department of Energy (DOE), Brookhaven National Laboratory conducts research in the physical, biomedical, and environmental sciences, as well as in energy technologies and national security. Brookhaven Lab also builds and operates major scientific facilities available to university, industry and government researchers. Brookhaven is operated and managed for DOE's Office of Science by Brookhaven Science Associates, a limited-liability company founded by the Research Foundation for the State University of New York on behalf of Stony Brook University, the largest academic user of Laboratory facilities, and Battelle, a nonprofit applied science and technology organization.
2013-11556 | INT/EXT | Newsroom










