Small Agents to Direct Improved MOF Structures
Scientists showed how metal-organic frameworks (MOFs) could be designed more effectively for applications such as catalysis or gas separation
November 30, 2020
 enlarge
enlarge
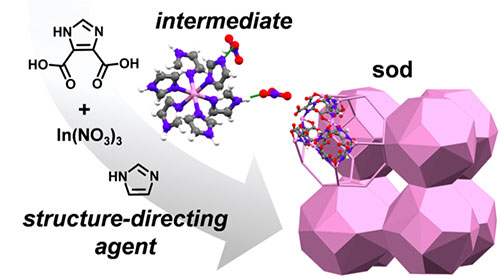
A structure-directing agent reacts with indium nitrate, In(NO3)3, to generate a key molecular intermediate that directs the formation of a 3-D sodalite (sod) structured framework material. Image credit: M. A. Sinnwell. J Am Chem Soc. 142, 17598−17606 (2020)
The Science
Scientists revealed that structure-directing agents (SDAs) influence structure formation during the synthesis of a metal organic framework (MOF).
The Impact
Customized MOFs are used for gas separation and catalysis; understanding how SDAs direct MOF formation will allow for a more rational and straightforward design of MOFs.
Summary
Gas separation applications, catalysts, and next-generation electronic devices can benefit from so-called metal-organic frameworks (MOFs). MOFs are complex molecular structures that are routinely built from small, flat, and relatively simple organic molecules – here referred to as structure-directing agents, or SDAs –, through a synthesis process. The effectiveness of MOFs depends on how the original atomic building blocks are arranged in 3D space during synthesis. However, based on the current knowledge of MOF formation, scientists have a hard time predicting the relationship between reaction parameters and outcomes.
In this study, a team of researchers investigated the role of SDAs during synthesis and discovered the formation of an SDA-based intermediate along the reaction pathway. This realization indicates that the role of the small SDA molecules goes beyond the current paradigm, that these additives are benign and balancing observers. Instead, they are reactive participants.
The team investigated an indium-based MOF system that is known to arrange into a variety of 3D architectures. They employed a combination of solid-state and liquid-state monitoring techniques to follow the synthesis process and witnessed the formation of an SDA-based intermediate along the reaction pathway. Among these monitoring techniques, the researchers used the X-ray Powder Diffraction (XPD) beamline at the National Synchrotron Light Source II (NSLS-II). NSLS-II is a U.S. Department of Energy (DOE) Office of Science User Facility located at DOE’s Brookhaven National Laboratory and offers access to a wide range of materials characterization tools to researcher from industry and academia.
Understanding the complex role structure-directing agents play in the reaction pathway will give researchers the tools needed to accomplish the synthesis of materials with predetermined and desirable structures.
Download the research summary slide
Contact
Michael A. Sinnwell
Pacific Northwest National Laboratory
michael.sinnwell@pnnl.gov
Praveen K. Thallapally
Pacific Northwest National Laboratory
praveen.thallapally@pnnl.gov
Publication
M.A. Sinnwell, Q.R.S. Miller, L. Palys, D. Barpaga, L. Liu, M. E. Bowden, Y. Han, S. Ghose, M. L. Sushko, H. T. Schaef, W. Xu, M. Nyman, P. K. Thallapally. Molecular Intermediate in the Directed Formation of a Zeolitic Metal-Organic Framework. Journal of American Chemical Society. 142, 17598−17606 (2020), DOI: 10.1021/jacs.0c07862.
Funding
This work was supported by the U.S. Department of Energy, Office of Science, Basic Energy Sciences, Materials Sciences and Engineering Division under Awards KC020105-FWP12152 (PNNL). Small-angle x-ray scattering work was done at OSU supported by the U.S. Department of Energy, Office of Science, Basic Energy Sciences, under Award DESC0010802. Structure determination was carried out using resources of the Advanced Photon Source, a U.S. Department of Energy (DOE) Office of Science User Facility operated for the DOE Office of Science by Argonne National Laboratory under Contract No. DE-AC02-06CH11357 from an XRD pattern collected at the beamline 28-ID-2 of the National Synchrotron Light Source II, a U.S. DOE Office of Science User Facility operated for the DOE Office of Science by Brookhaven National Laboratory under Contract No. DESC0012704. In situ XRD efforts were supported by the U.S. Department of Energy, Office of Science, Office of Basic Energy Sciences, Chemical Sciences, Geosciences, and Biosciences Division through its Geosciences program at PNNL. The microscopy portion of this research was performed in the Environmental Molecular Sciences Laboratory, a national scientific user facility sponsored by the Department of Energy’s Office of Biological and Environmental Research and located at PNNL. PNNL is a multiprogram national laboratory operated for the U.S. Department of Energy by Battelle Memorial Institute under Contract DE-AC05-76RL01830.
2020-18793 | INT/EXT | Newsroom









