CFN User Spotlight: Nik Singh Seeks Better Battery Materials
interview with a CFN user
January 15, 2020
 enlarge
enlarge
Nik Singh, a senior scientist at the Toyota Research Institute of North America, studies post-lithium-ion battery systems.
Since 2011, Nikhilendra (Nik) Singh has been a senior scientist in the Materials Research Department at the Toyota Research Institute of North America (TRINA)—a division of Toyota Motor North America Research and Development (TMNA R&D)—based in Ann Arbor, MI. His quest to find alternatives to lithium (Li)-ion batteries has brought him to the Center for Functional Nanomaterials (CFN), a U.S. Department of Energy (DOE) Office of Science User Facility at Brookhaven National Laboratory. He holds a PhD in chemistry from Purdue University and a bachelor’s degrees in chemistry and environmental science from Muskingum University.
What are the missions of TMNA R&D and TRINA?
TMNA R&D aims to bring the brightest minds in automotive exploration together to redefine the next generation of mobility. Our goal is to leverage the power of fully connected vehicles to enable people to move safely and comfortably.
Based in Ann Arbor, MI, TMNA R&D—founded in 1977 and formerly known as the Toyota Technical Center—is the R&D arm of Toyota Motor North America. Engineers lead the development of eight Toyota models in North America, as well as prototype and concept development, including the Toyota Research Institute’s Platform 3 and 4 automated driving test vehicles. The team also leads North American tuning and development for advanced powertrains and alternative fuel vehicles, and spearheads advanced technological innovations related to materials and electronics research.
At TMNA R&D’s Gardena, CA, campus, engineers and researchers support the implementation of hydrogen fuel cell electric vehicles (FCEVs) in North America. In addition, TMNA R&D manages the Toyota Arizona Proving Grounds and the Collaborative Safety Research Center in Ann Arbor, MI.
TRINA was founded more than 10 years ago to conduct basic and applied research on new technology to support Toyota’s future business in mobility. TRINA’s role in the pursuit of sustainable mobility addresses four key priorities: advanced technologies, the urban environment, energy, and partnerships with government and academia. The specific research topics of interest include advanced batteries, catalysts, fuel cells, thermal management, power electronics, vehicle sensors, human-machine interfaces, smart materials, informatics, and sociotechnical systems.
TRINA’s research spans many different technologies. What does your personal research focus on?
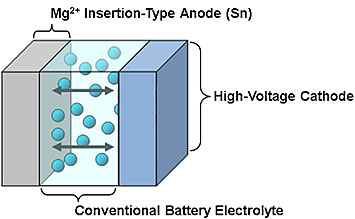
My personal research focus is post-Li-ion battery systems such as multivalent ion (e.g., magnesium (Mg), zinc (Zn), calcium (Ca), and aluminum (Al)), Li-air, Li-metal-based, and all-solid-state batteries. We continue to develop next-generation batteries past Li-ion, as we believe that battery diversification is important for future electric vehicle (EV) and plug-in hybrid vehicle (PHV) applications.
For my research, I work as part of a team at Toyota, both locally and internationally, partnering with battery research groups in the United States and Japan.
 enlarge
enlarge
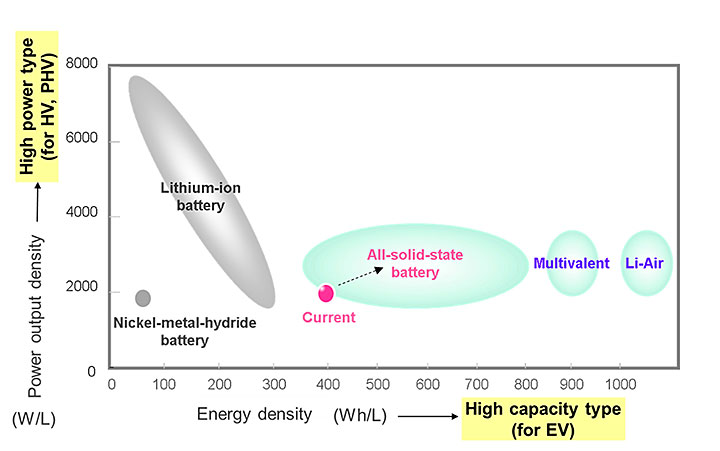
A graph illustrating some of the battery technologies under consideration at Toyota for current and future hybrid vehicle (HV), plug-in hybrid vehicle (PHV), and electric vehicle (EV) applications.
Explain the different components of a battery and how a battery works. Which part(s) of the battery do you target for your studies?
In general, batteries are composed of three primary components: a cathode, or the positive electrode of the battery; an anode, or the negative electrode of battery; and an electrolyte, or the material located between the anode and cathode that makes the battery work by promoting movement of ions or charge from the cathode to the anode and vice-versa.
A battery is essentially a device capable of storing electrical energy in the form of chemical energy. The chemical reactions, which occur at the electrodes (cathode and anode), create a flow of electricity when a load is added to the battery.
At TRINA, we research all aspects of batteries. However, I specifically study anode and electrolyte components and their interfaces.
The Toyota Prius—a Latin word meaning “coming before”—was the first mass-produced hybrid electric vehicle, launched in 1997 in Japan and released worldwide in 2000. Several all-electric, hybrid electric, and plug-in hybrid electric vehicles have since entered the market, but they have not been adopted on a widespread scale. What are some of the reasons why these vehicles have not become mainstream?
The decision to buy one vehicle over another one is an extremely complex one that considers factors such as cost, performance, style, and utility. The environmental impact of a vehicle is certainly an item on that list, but it isn’t the top concern for a lot of consumers. Also, sometimes it just takes time for a new technology to fully make its way into the market. Cars have been around for a long time, and the gasoline engine is a well-known technology that people are comfortable with. But, the greatest impact is likely the relatively low cost of gasoline over the past decade since the beginning of the fracking revolution. The sales of hybrid and other electrified powertrains correlate very strongly with gas prices, historically.
Toyota has sold more than 14 million hybrid electric vehicles (HEVs) globally, and more than 3.6 million in North America. Although industry demand for cars versus trucks has shifted, we are seeing signs that consumers agree that there remains a place for hybrid electrics, with an increase in demand for vehicles like the RAV4 Hybrid. In 2018, we sold 48,124 RAV4 Hybrids, making it our second most popular hybrid behind the Prius family, which sold 87,590. With the inevitable increases in fuel prices in the future, the hybrid electric powertrain remains a part of our core powertrain strategy and will ultimately bring more buyers into the hybrid space.
Toyota will continue to benefit from a balanced portfolio that meets the needs of all our customers. In 2018, Toyota hybrid sales (excluding Prius Prime and Mirai) accounted for nearly 44 percent of overall U.S. hybrid sales. In 2018, more than nine percent of our sales were made up of HEVs, plug-in electric vehicles (PEVs) and FCEVs. We anticipate that figure to grow to more than 15 percent by 2020.
What will it take to make more cost-effective and energy-efficient electric vehicles? How far out are we from those advances?
Like many products, for energy-efficient electric vehicles to become more ubiquitous, many technologies need to come together. In this case, we need higher-energy-density batteries (to improve range), more compact power electronics, improvements in motor technology, and a charging mechanism and infrastructure that customers will accept. All of these technologies also need to get cheaper. Fortunately, there are a lot of smart people all over the world chipping away at these problems every day. As for when this will all come to fruition, that is hard to predict. Some analysts think the electric revolution is just a decade away. Others believe it might take 30 years or more.
As evidenced by the number of electrified vehicles we have sold, the efficiency is already being experienced by a wide variety of consumers. And, as technology continues to improve and advance, we expect greater efficiency to proliferate to all of our product segments. In fact, by 2025, our global goal is for an electrified option (hybrid, plug-in hybrid, hydrogen fuel cell electric, or battery electric) to be available on all Toyota and Lexus models. By 2025, approximately 50 percent (5.5 million) of our new vehicles sold per year globally will be electrified in some form.
Li-ion batteries have dominated the electric vehicle space, but they have their limitations. What are some of the other battery materials of interest to Toyota?
It is believed that energy storage systems will play a pivotal role in future energy production, grid storage, and transportation demands. Since the commercialization of Li-ion batteries in the 1990s, incremental improvements to the performance of Li-ion batteries via alterations to battery energy density, capacity, and cycle life have paved the pathway to their use in electric vehicles today. For transportation applications, improvements in driving range and space (not occupied by batteries) within electric vehicles remain of paramount focus. From a battery materials perspective, one way to improve both driving range and space is to use battery materials that exhibit improved volumetric energy density in comparison to commercial Li-ion batteries. Prime candidates for such improved volumetric energy densities are alternative multivalent ion systems, such as the ones I mentioned before.
A particularly promising avenue among these systems has been batteries based on Mg metal, which exhibits a high negative reduction potential (resulting in a large voltage window), lower cost based on its relative abundance in comparison to Li, potential battery safety advantages due to the absence of dendrite growth during battery operation (dendrites are spiny metal projections that grow from an electrode’s surface and can short circuit the battery and in some cases cause fires), and a significant advantage in volumetric energy density over existing Li-ion battery graphite anodes. To date, the best performing Mg battery system remains the one demonstrated by Aurbach et. Al. in 2000, which coupled a “Chevrel phase” cathode and an Mg anode.
In order to develop post-Li-ion energy storage systems, you need tools to synthesize and characterize the materials. What capabilities exist at Toyota?
Toyota’s global research activity and investment is huge. Here in North America, our research division has state-of-the-art research labs that enable us to fabricate, analyze, and evaluate new devices and materials. In addition to our in-house work, our research team is currently engaged in dozens of collaborative research projects throughout North America to evaluate and develop the most cutting-edge technologies.
How did you come to conduct research at the CFN?
I came to learn of the CFN via interactions with Eric Stach, former group leader of Electron Microscopy at the CFN and now a professor of materials science and engineering at the University of Pennsylvania. I met Eric while I was doing my PhD at Purdue, where he was a professor at the time. We interacted over microscopy-related work and classes, and I reached out to him again once he was at Brookhaven Lab. Because of our interactions with Eric and the great instrumentation available at the CFN for ex situ and in situ transmission electron microscopy (TEM), we decided to submit a user proposal.
Here at TRINA, we do not have TEM capabilities, especially those related to in situ TEM, wherein the CFN Electron Microscopy Group excels. At the CFN, we primarily used the FEI Talos for both our ex situ and in situ TEM experiments, and the focused-ion beam (FIB) scanning electron microscope (SEM) in the cleanroom for certain sample preparation.
 enlarge
enlarge
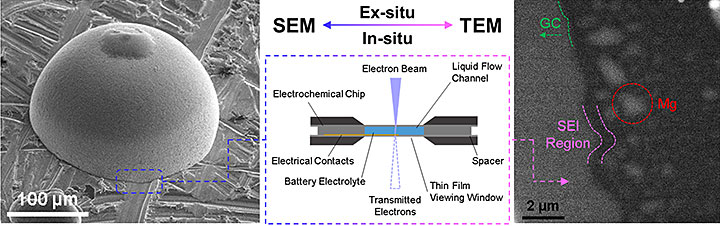
A representative image highlighting the interaction between ex situ and in situ microscopy. From ACS Applied Energy Materials, 2018, 1, 9, 4651–4661.
What information about your battery materials does TEM provide?
TEM is a very versatile and powerful tool for materials science. Very simply, a TEM operates by focusing and transmitting a high-energy electron beam through a very thin sample. As a result of the interactions between the electrons from the beam and the atoms of the sample, one can observe and image features like crystal structure, grain boundaries, and dislocations within the sample. TEMs can be outfitted with very powerful analytical tools such as energy dispersive X-ray spectroscopy (EDS) and electron energy loss spectroscopy (EELS) systems, which can provide chemical analysis of the sample alongside the imaging analysis. Chemical composition and identification information are critical to investigations of new material properties.
 enlarge
enlarge
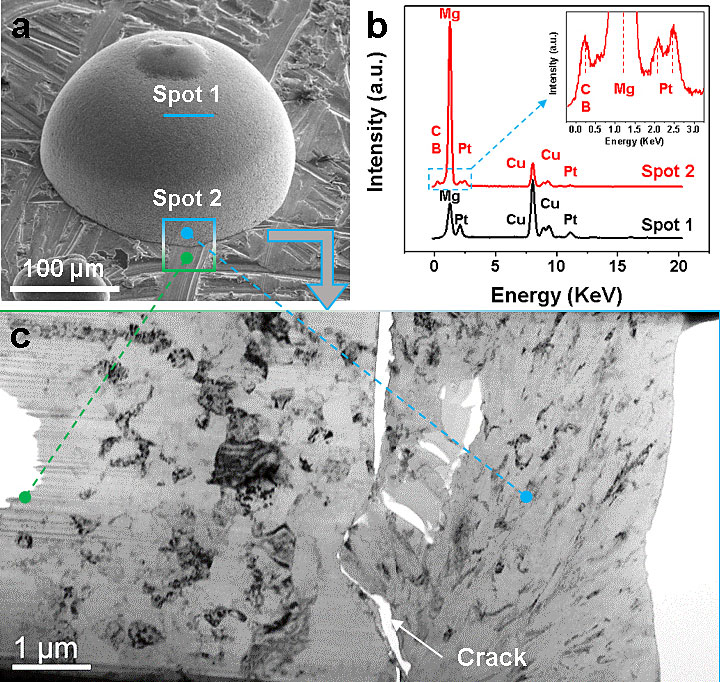
(a) An SEM image highlighting the representative areas of Spot 1 and Spot 2, from where FIB samples were processed for energy-dispersive x-ray spectroscopy (EDS) and TEM analysis. (b) An EDS spectra for the FIB samples obtained from Spot 1 and Spot 2 in (a). (c) A TEM image of the FIB sample obtained from Spot 2 in (a), displaying a variation in sample morphology/texture through the interface of deposition from the Mg foil substrate, extending into the newly deposited Mg from MMC/G4 (an electrolyte system). From ACS Applied Energy Materials, 2018, 1, 9, 4651–4661.
We can conduct in situ experiments on battery materials at the CFN by using specially designed in situ TEM holders made by Hummingbird Scientific (a company that Eric Stach co-founded). These holders allow us to recreate model experiments that imitate processes and mechanisms happening in real batteries while allowing us to image and chemically analyze them in real time. Through such studies, we can observe the growth of layers at material interfaces and the composition and defects of these layers—all of which can strongly govern the performance of battery materials. For our work at the CFN, we used an in situ liquid electrochemistry holder, which allowed us to observe the deposition of materials like Li and Mg from liquid electrolytes within the TEM. We were able to identify specific characteristics about the interfaces formed between these materials and the electrolytes from which they were electrochemically deposited.
 enlarge
enlarge
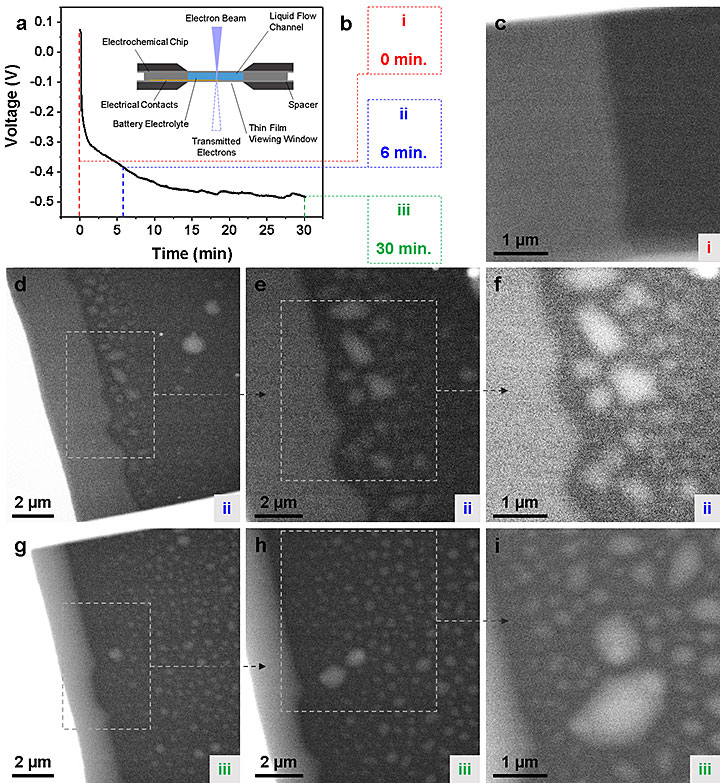
(a) A potentiometric response captured from the operando scanning TEM (STEM) liquid electrochemistry holder upon the application of 1 mA cm−2 (at 0.5 mA·h) with a schematic representation of the holder (inset). (b) Relevant information regarding data collected during the experiment. (c) An operando TEM image taken at 0 min (0.0 mA·h) of Mg deposition time in (a). (d−f) Operando TEM images taken at 6 min (0.1 mA·h) of Mg deposition time in (a). (g−i) Operando TEM images taken at 30 min (0.5 mA·h) of Mg deposition time in (a). From ACS Applied Energy Materials, 2018, 1, 9, 4651–4661.
How did your interactions with Eric—and any other CFN staff members—impact your experience as a CFN user?
 enlarge
enlarge
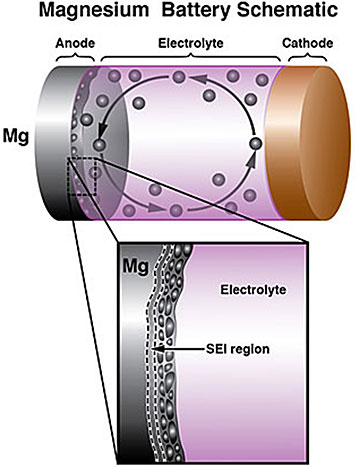
Schematic of a rechargeable battery with a magnesium (Mg) anode (top). A close-up of the Mg anode/electrolyte interface, showing the solid electrolyte interphase and formation of Mg nanocrystals during battery operation (bottom).
In addition to Eric, we worked with Huolin Xin, Kim Kisslinger, and Jing Li. Eric was a co-principal investigator on all of the project work conducted at the CFN and was critical in planning our experiments to extract the information we were seeking—the mechanism by which specific Mg battery electrolyte systems deposit Mg during battery operation. This planning in turn allowed both Eric and Huolin to best operate the microscope for our experiments to continuously collect good relevant data. Kim and Jing were very involved from a standpoint of support with microscope operation, sample preparation, and both ex situ and in situ data collection.
Complementary capabilities for battery material characterization exist at Brookhaven’s National Synchrotron Light Source II (NSLS-II)—another DOE Office of Science User Facility. Could your research benefit from these capabilities?
We are always interested in being able to use advanced analysis techniques to further our understanding of the materials and systems we research. TEM is one such example. The capabilities of a light source like NSLS-II would be great to add to the analysis realm of our research work. One of our major focus areas with analysis techniques has always been to find complementary techniques that bridge length scales. TEM provides down to atomic resolution but is unable to realistically analyze materials larger than micron size, while x-ray synchrotron techniques can image from nanometer to millimeter scales. Such complementary information allows us to understand focused phenomena in a material at very high resolution and tie them to phenomena occurring at much larger length scales—all the way up to a device itself.
In April 2019, Toyota announced that it will offer free access to nearly 24,000 of its hybrid vehicle patents through 2030. How do you think this open access will impact the development and adoption of electric vehicles?
We made this decision based on a desire to accelerate the proliferation of electrified vehicles in order to contribute to reductions in carbon dioxide (CO2) emissions. Toyota has long considered environmental issues a top-priority management issue and has undertaken development and sales of HEVs and FCEVs from an early stage.
Toyota has set challenging targets for reducing CO2 emissions. On the basis of a strong belief that environment-friendly vehicles will contribute to the environment only if they come into widespread use, we are taking proactive measures to promote the development and sales of electrified vehicles, centered on our core vehicle electrification technologies, such as the motors, batteries, and power conditioning units (PCUs) (inverters) cultivated through HEV development. In addition, we are striving to share technologies and systems in accordance with our stance that environmental technologies should not be monopolized.
Toyota will provide fee-based technical support to other manufacturers developing and selling electrified vehicles when they use Toyota's motors, batteries, PCUs, electronic control units, and other vehicle electrification system technologies as part of their powertrain systems.
It is an exciting time for battery R&D. What do you enjoy most about being at the cutting edge of this field?
Research to me is exciting because it allows me to creatively solve problems that can directly impact our future. Being able to one day make an impact on our scientific and general society would be quite an accomplishment.
Materials science spans many different disciplines. What led you to study battery materials, and when did your interest in science and more specifically chemistry first begin?
My interest in science certainly began in my childhood. My parents were both significantly involved with academia, fostering an environment where I grew up exposed to multiple facets of science. My general interest in chemistry began during my undergraduate days, where I chose chemistry as a collaborative track to environmental science.
During my undergraduate days, my simultaneous interests in chemistry and environmental science led me to consider ways to bridge the two subjects in sustainable ways. One obvious path here was the energy field. Working on battery materials gave me an avenue to research and progress energy storage, which can provide sustainable alternatives to traditional technologies.
Interested in becoming a CFN user? Submit a proposal through the online proposal system: https://cfnproposals.bnl.gov. If you have questions about using CFN facilities or partnering with CFN scientists, please contact CFN Assistant Director for Strategic Partnerships Priscilla Antunez at (631) 344-6186 or pantunez@bnl.gov.
Brookhaven National Laboratory is supported by the Office of Science of the U.S. Department of Energy. The Office of Science is the single largest supporter of basic research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time. For more information, please visit http://energy.gov/science.
Follow @BrookhavenLab on Twitter or find us on Facebook.
2020-17018 | INT/EXT | Newsroom