Tripod Nanocatalysts Support Faster Fuel Cell Reactions
February 28, 2018
What is the scientific achievement?
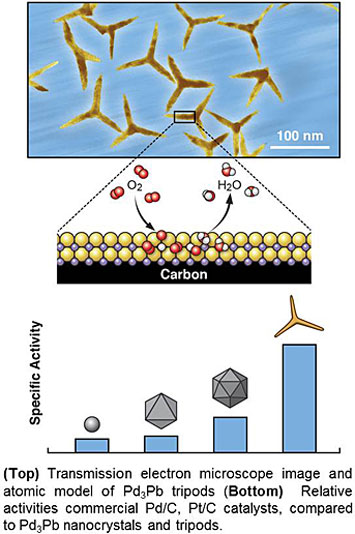
Highly-efficient catalysts for the oxygen reduction reaction are important for fuel cell performance. Platinum-based catalysts have shown the best performance, but it is desirable to replace these due to high cost and the relative scarcity of Pt. Collaborators from Soochow Univ., Peking Univ., Hong Kong Polytechnic Univ., and CFN designed and characterized palladium-lead (Pd3Pb) catalysts shaped like tripods, which show improved catalytic activity for the oxygen reduction reaction compared to commercial Pt/C and Pd/C catalysts, and maintain high performance over 20,000 cycles.
Why does this achievement matter?
This work demonstrates the promise of rational design of high-performance catalysts through appropriate control of nanomaterial facets and electronic structures.
What are the details?
Highly efficient oxygen reduction reaction (ORR) catalysts are a key factor for developing high-performance fuel cells and metal-air batteries. Palladium (Pd) is a class of ORR catalyst with potential for replacing more expensive and rare platinum (Pt); however it typically has much lower ORR electrocatalytic activity compared to Pt. In this work, collaborators from Soochow University, Peking University, Hong Kong Polytechnic University, and CFN report a class of structurally ordered Pd3Pb tripods with predominantly {110} exposed crystal facets, that shows extremely high activity and stability for ORR in alkaline conditions. First principles calculations suggest that strong charge exchange between Pd-4d and Pb-(sp) orbitals in the {110} facets results in a Pd-Pb local bonding unit with an orbital configuration similar to Pt. As a consequence, Pd3Pb tripods exhibit much higher specific and mass activities for ORR than either commercial Pt/C and commercial Pd/C catalysts. The Pd3Pb tripods are durable for ORR, sustaining over 20,000 potential cycles under alkaline condition with negligible structural and compositional changes.
CFN Capabilities
CFN Electron Microscopy facilities were used for characterizing the catalyst structure.
Publication Reference
Lingzheng Bu,1 Qi Shao,1 Yecan Pi,1 Jianlin Yao,1 Mingchuan Luo,2,3 Jianping Lang,1 Sooyeon Hwang,4 Huolin Xin,4 Bolong Huang,5* Jun Guo,6 Dong Su,4* Shaojun Guo,2,3* and Xiaoqing Huang1*, Coupled s-p-d Exchange in Facet-Controlled Pd3Pb Tripods Enhances Oxygen Reduction Catalysis, Chem, XX, XXXX
Acknowledgement of Support
This work was financially supported by the Ministry of Science and Technology (2016YFA0204100, 2017YFA0208200), the National Natural Science Foundation of China (21571135 and 51671003), the National Key Research and Development Program of China (No. 2016YFB0100201), Young Thousand Talented Program, the start-up supports from Soochow University, and the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD). Part of the electron microscopy work was performed at the Center for Functional Nanomaterials, Brookhaven National Laboratory, which is supported by the U.S. Department of Energy (DOE), Office of Basic Energy Science, under contract DE-SC0012704.
2018-12816 | INT/EXT | Newsroom