Understanding 'Strain-Free' Energy Storage in Lithium-ion Batteries
August 31, 2018
What is the scientific achievement?
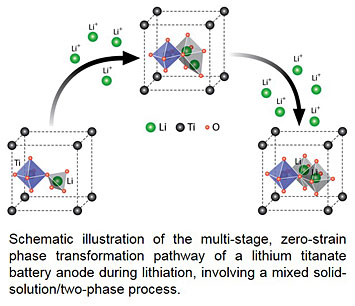
Scientists from Stony Brook University, BNL, and the CFN identified key features contained in in-situ X-ray absorption spectra (XAS) that reveal the complex, multi-stage phase transformation pathway by which lithium titanate battery anodes store lithium without undergoing strain. First-principles simulations of the material were key to understanding features in the XAS fingerprints.
Why does this achievement matter?
The insights into the complex material phase transformation gained from this study are essential for understanding battery electrode performance and rational design of new zero-strain materials, which are preferred for long-lasting rechargeable batteries. The technique of linking in-situ XAS fingerprints to material structural changes can be broadly applied to other lithium-ion battery electrodes.
What are the details?
Zero-strain electrodes, such as spinel lithium titanate (Li4/3Ti5/3O4), are appealing for battery applications due to their negligible volume change and extraordinary stability upon repeated charge/discharge cycles. However, this same property makes it challenging to probe their structural changes during the electrochemical reaction. In this work, we report in situ studies of lithiation-driven structural transformations in Li4/3Ti5/3O4 via a combination of X-ray absorption spectroscopy with ab initio calculations. Based on excellent agreement between computational and experimental spectra of the Ti K-edge, we identified key spectral features as fingerprints for quantitative assessment of structural evolution at different length scales. Results from this study indicate that, despite small variation in the crystal lattice, pronounced structural transformations occur during lithiation in Li4/3Ti5/3O4, both locally and globally, giving rise to a multi-stage kinetic process involving mixed solid-solution / two-phase transformations over a wide range of Li concentrations. This work highlights the unique capability of combining in situ core-level spectroscopy and calculations for real time probing of Li-ion intercalation in zero-strain and other types of electrodes. The findings on structure-determined electrochemical properties in Li4/3Ti5/3O4 may provide insights into designing new zero-strain electrode materials for long-life batteries.
CFN Capabilities:
The CFN Theory and Computation facility was used to model X-ray absorption spectra.
Publication Reference
Wei Zhang, Mehmet Topsakal, Christina Cama, Christopher J. Pelliccione, Hu Zhao, Steven Ehrlich, Lijun Wu, Yimei Zhu, Anatoly I. Frenkel, Kenneth J. Takeuchi, Esther S. Takeuchi, Amy C. Marschilok, Deyu Lu, Feng Wang, Multi-Stage Structural Transformations in Zero-Strain Lithium Titanate Unveiled by in Situ X-ray Absorption Fingerprints, Journal of the American Chemical Society 139, 16591 (2017).
DOI: 10.1021/jacs.7b07628
https://pubs.acs.org/doi/abs/10.1021/jacs.7b07628
Acknowledgement of Support
This work was supported by the Center for Mesoscale Transport Properties, an Energy Frontier Research Center supported by the U.S. Department of Energy, Office of Science, Basic Energy Sciences, under award no. DE-SC0012673. The effort on calculations by M.T. was supported by LDRD Project at BNL (No. 16-039). The effort on interpretation of X-ray absorption by A.I.F. was supported by the U.S. Department of Energy grant no. DE-FG02-03ER15476. The effort on electron microscopy measurements by L.W. and Y.Z. was supported by the U.S. Department of Energy, Office of Basic Energy Science, Division of Materials Science and Engineering, under Contract No. DE-SC0012704. The use of the National Synchrotron Light Source was also supported by the U.S. Department of Energy, Office of Science, Basic Energy Sciences, under Contract No. DE-SC0012704. This research used resources of the Center for Functional Nanomaterials, which is a U.S. DOE Office of Science Facility, at Brookhaven National Laboratory under Contract No. DE-SC0012704. This research used resources of the National Energy Research Scientific Computing Center, a U.S. DOE Office of Science User Facility supported by the Office of Science of the U.S. Department of Energy under Contract No. DE-AC02-05CH11231.
2018-13119 | INT/EXT | Newsroom