Chemical Modification of Self-Assembled Block Copolymers for Area-Selective Formation of Metal Oxide Nanostructures
February 13, 2013
What is the Scientific Achievement?
We have chemically modified self-assembled polystyrene-PMMA block copolymer thin films by ultraviolet (UV) light to enhance their ability to localize selectively weakly interacting organometallic precursors. This enabled the formation of metal oxide (zinc oxide, titania, and alumina) nanopatterns and their inverses using the same starting block copolymer template. Metal oxide nanostructures exhibit properties and great potential for device and energy applications. Our results expand the functionality of PS-PMMA block copolymer templates for more precise control of their nanometer-scale morphologies. Using near-edge x-ray absorption fine structure and Fourier transform infrared spectroscopies, we measured UV-induced formation of oxygen-containing functional groups within the block copolymers, increasing their Lewis base–acid interaction with organometallic precursors and enhancing the area-selective metal oxide formation. These results suggest ways for developing and identifying new block copolymers that extend the range of metal oxide nanostructures that can be synthesized using them as templates.
Why Does This Matter?
Block copolymers are valuable templates for nanomaterials synthesis, but their use has been limited by the organometallic precursors that can bind to them. Photo-chemical modification of a common block copolymer has enhanced its affinity for metals and extended the technique to include more metal oxides of importance for energy applications.
What Are The Details?
- Ultraviolet irradiation of block copolymer thin films enables area-selective synthesis of a variety of metal oxide nanostructures from weakly interacting organometallic precursors.
- Synchrotron x-ray absorption and Fourier transform infrared spectroscopies reveal the formation of oxygen-containing functional groups (Lewis bases) in the copolymer, which attract organometallic precursors (Lewis acids).
- The approach provides a method for generating either metal oxide nanoscale patterns or their inverses using the same starting block copolymer film.
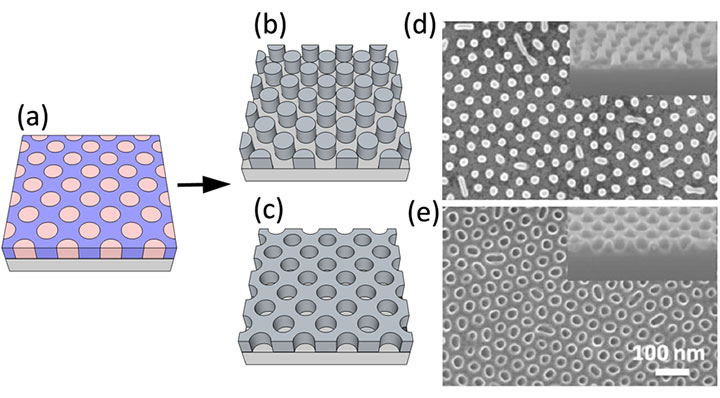
Schematic representations of: (a) self-assembled block copolymer thin film; (b) metal oxide nanopillars that resulted from infiltration synthesis on the as-assembled template; and (c) metal oxide nanoholes (i.e., inverse pattern of (b)), synthesized using the ultraviolet-light-irradiated template. High-resolution scanning electron micrographs of (d) nanopillar and (e) nanohole patterns.
Publication Reference
Chemically Enhancing Block Copolymers for Block-Selective Synthesis of Self-Assembled Metal Oxide Nanostructures
Jovan Kamcev,† David S. Germack,‡ Dmytro Nykypanchuk,† Robert B. Grubbs,† Chang-Yong Nam,† and Charles T. Black,†
†Center for Functional Nanomaterials, Brookhaven National Laboratory, Upton, New York 11973, United States
‡Condensed Matter Physics and Materials Science Department, Brookhaven National Laboratory, Upton, New York 11973, United States
ACS Nano 7, 339 (2013)
Acknowledgment of Support
This research is supported by the U.S. Department of Energy, Basic Energy Sciences, at the Center for Functional Nanomaterials (J.K., D.N., R.B.G., C.-Y.N., and C.T.B.) and the Materials Sciences and Engineering Division (D.S.G.) (Contract No. DE AC02 98CH10886). This work was partially supported by the Energy Laboratory Research and Development Initiative at Brookhaven National Laboratory.
2013-3702 | INT/EXT | Newsroom









