Resonant Energy Transfer from Quantum Dots to Graphene
May 21, 2014
What Is The Scientific Achievement?
 enlarge
enlarge
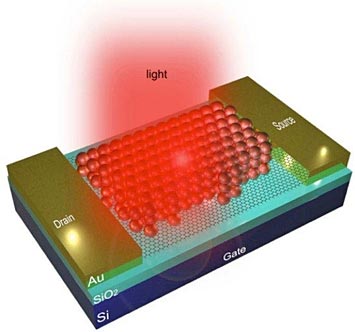
Schematic of a quantum dot-graphene nano-photonic device, as described in this research project.
Semiconductor quantum dots (QDs) are nanoscale semiconductors that exhibit size dependent physical properties. For example, the color (wavelength) of light that they absorb changes dramatically as the diameter decreases. Graphene is an atomically thick sheet of carbon atoms, arranged in a hexagonal lattice pattern. In this work, QDs have been combined with graphene to develop nanoscale photonic devices that can dramatically improve our ability to detect light. Quantum dots can absorb light and transfer it to graphene, but the efficiency of the transfer depends on how far the QDs and the graphene are separated from each other. This study demonstrated that the thickness of the organic molecule layer that typically surrounds the QDs is crucial in attaining sufficiently high efficiency of this light/energy transfer into the graphene. In other works, the thinner the organic layer, the better. This transfer can be further optimized by engineering the interface between the two nanomaterials, specifically optimizing the thickness of the organic capping molecules on the quantum dots. Based on this work, further improvement of the performance of these nano-photonic devices can be expected.
Why Does This Matter?
 enlarge
enlarge
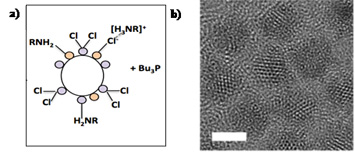
a) Schematic of a chloride-terminated CdSe quantum dot. b) A high resolution transmission electron microscopy image of such quantum dots.
Commercial cadmium selenide (CdSe) quantum dots have long insulating organic ligands that prevent their utilization in energy and charge transfer applications for which short distances between the QDs and other materials are critical. Short, chlorine ligands that passivated CdSe QDs are an intriguing alternative material to enhance the interaction with materials into which charge carriers, such as electrons, can easily conduct. Graphene is such a material. The combination of CdSe quantum dots and graphene could hold the key to the development and implementation of nanoscale materials systems in flexible electronics and photodetectors.
 enlarge
enlarge
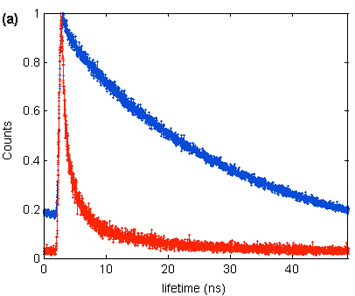
Photoluminescence lifetime decay of isolated quantum dots on glass (blue) and graphene (red) demonstrate efficient energy transfer between the quantum dots and graphene.
What Are The Details?
- CFN Capabilities: The Advanced Optical Microscopy Facility measured the time-resolved photoluminescence from isolated CdSe quantum dots deposited on graphene.
- The team discovered that short, chloride-capped CdSe quantum dots, deposited on chemical-vapor-deposited, monolayer layer graphene, exhibited highly efficient energy transfer to the graphene with a 4x observed reduction in the excitonic lifetime. This demonstrated significant near-field coupling between quantum dots and the graphene.
Publication Reference
Time-resolved energy transfer from single chloride-terminated nanocrystals to graphene
Obafunso A. Ajayi1, Nicholas C. Anderson2, Mircea Cotlet3, Nicholas Petrone4, Tingyi Gu1, Felice Gesuele1, Jonathan S. Owen2, James Hone4, Chee Wei Wong1
1 Optical Nanostructures Laboratory, Center for Integrated Science and Engineering, Solid-State Science and Engineering, and Mechanical Engineering, Columbia University, New York, NY 10027
2 Department of Chemistry, Columbia University, New York, NY 10027
3 Brookhaven National Laboratory, Upton, New York, NY 11973
4 Department of Mechanical Engineering, Columbia University, New York, NY 10027
Applied Physics Letters 104, 171101 (2014).
Acknowledgment of Support
This work was supported as part of the Center for Redefining Photovoltaic Efficiency through Molecule Scale Control, an Energy Frontier Research Center funded by the U.S. Department of Energy, Office of Science, Basic Energy Sciences under Award DE-SC0001085. O.A.A. acknowledges a Graduate Research Fellowship through the National Science Foundation. Research carried out in part at the Center for Functional Nanomaterials, Brookhaven National Laboratory, which is supported by the U.S. Department of Energy, Office of Basic Energy Sciences, under Contract No. DE-AC02-98CH10886.
2014-4906 | INT/EXT | Newsroom









