Peering into How Rechargeable Lithium Ion Batteries Function
March 18, 2015
What Is The Scientific Achievement?
 enlarge
enlarge
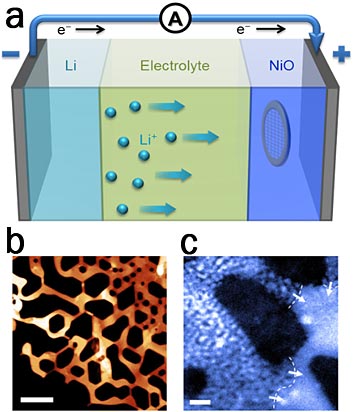
a) Schematic of a rechargeable lithium ion battery cell with a TEM grid, which is used for electron microscopy analysis, submerged inside the electrode. (b-c) scanning transmission electron microscopy images of a NiO electrode when b) pristine (scale bar 100nm) and when c) half-reacted (scale bar 10nm).
Nanoparticle electrodes in lithium-ion batteries have both near-surface and interior contributions to their redox capacity, each with distinct rate capabilities. Using combined electron microscopy, synchrotron X-ray methods and ab initio calculations, we have investigated the lithiation pathways that occur in NiO electrodes. We find that the near-surface electroactive (Ni2+→Ni0) sites saturated very quickly, and then encounter unexpected difficulty in propagating the phase transition into the electrode (referred to as a “shrinking-core” mode). However, the interior capacity for Ni2+→Ni0 can be accessed efficiently following the nucleation of lithiation fingers, which propagate into the sample bulk, but only after a certain incubation time. Our microstructural observations of the transition from a slow shrinking-core mode to a faster lithiation finger mode corroborate with synchrotron characterization of large-format batteries, and can be rationalized by stress effects on transport at high-rate discharge. The finite incubation time of the lithiation fingers sets the intrinsic limitation for the rate capability (and thus the power) of NiO for electrochemical energy storage devices. The present work unravels the link between the nanoscale reaction pathways and the C-rate-dependent capacity loss, and provides guidance for the further design of battery materials that favors high C-rate charging.
Why Does This Matter?
Understanding the link between nanoscale reaction pathways and the resulting electric properties of lithium ion batteries can provide considerable information about how to improve the overall design and longevity of these rechargeable batteries.
What Are The Details?
- CFN Capabilities: CFN’s Electron Microscopy Facilities were used for atomic scale imaging, spectroscopy, and tomography during the in-situ lithiation process.
Publication Reference
Transitions from Near-Surface to Interior Redox upon Lithiation in Conversion Electrode Materials
Kai He†,¶, Huolin L. Xin†,¶, Kejie Zhao‡, Xiqian Yu§, Dennis Nordlund||, Tsu-Chien Weng||, Jing Li†,⊥, Yi Jiang#, Christopher A. Cadigan∇, Ryan M. Richards∇, Marca M. Doeffâ—‹,Xiao-Qing Yang§, Eric A. Stach†, Ju Li‡,*, Feng Lin∇,â—‹,*, and Dong Su†,⊥,*
†Center for Functional Nanomaterials, Brookhaven National Laboratory, Upton, New York 11973, USA.
‡Department of Materials Science and Engineering, Massachusetts Institute of Technology, Cambridge, Massachusetts 02139, USA.
§Chemistry Department, Brookhaven National Laboratory, Upton, New York 11973, USA.
||Stanford Synchrotron Radiation Lightsource, SLAC National Accelerator Laboratory, Menlo Park, California 94025, USA.
⊥Department of Materials Science and Engineering, Stony Brook University, Stony Brook, New York 11794, USA.
#Department of Physics, Cornell University, Ithaca, New York 14853, USA.
∇Department of Chemistry and Geochemistry, Materials Science Program, Colorado School of Mines, Golden, Colorado 80401, USA.
â—‹Environmental Energy Technologies Division, Lawrence Berkeley National Laboratory, Berkeley, California 94720, USA.
Nano Letters Nano Lett. 15, 1437-1444 (2015).
Acknowledgment of Support
The electron microscopy work (S/TEM, EELS and tomography) was carried out at the Center for Functional Nanomaterials, Brookhaven National Laboratory, which is supported by the U.S. Department of Energy (DOE), Office of Basic Energy Sciences, under Contract No. DE-AC02-98CH10886 and DE-SC-00112704. The authors acknowledge the technical support from Dr. Steven N. Ehrlich at Beamline X18A of National Synchrotron Light Source at Brookhaven National Laboratory, as well as the support from Beamline 9-BM-B at APS. X.Y. and X.-Q.Y. were supported by the U.S. DOE, the Assistant Secretary for Energy Efficiency and Renewable Energy, Office of Vehicle Technologies under Contract Number DE-AC02-98CH10886 and DE-SC-00112704. The synchrotron X-ray work was partially carried out at the Stanford Synchrotron Radiation Lightsource, SLAC National Accelerator Laboratory, supported by the U.S. DOE under Contract No. DE-AC02-76SF00515. Y.J. is supported by DOE Grant DE-FG02-11ER16210. Ju L. acknowledges support by NSF DMR-1240933 and DMR-1120901. Computational time on the Extreme Science and Engineering Discovery Environment (XSEDE) under the grant number TG-DMR130038 is gratefully acknowledged.
2015-5595 | INT/EXT | Newsroom









