Efficient Conversion of CO2 into Methanol Using Nanocatalysts
November 6, 2015
What Is The Scientific Achievement?
 enlarge
enlarge
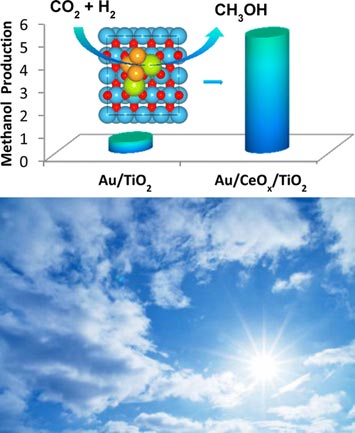
Top) Carbon dioxide molecules, the main component of greenhouse emissions, are converted into valuable methanol at the interface between Au nanoparticles and a mixed oxide (CeOx/TiO2) support under unprecedented low pressures of hydrogen. Bottom) This could lead to breakthroughs on greenhouse gas emission remediation.
The capture and recycling of carbon dioxide (CO2) not only prevents its emission into the atmosphere but also can lead to the production of valuable chemicals, such as methanol. This study demonstrates that gold (Au) nanoparticles anchored and stabilized on a substrate comprised of a mixture of ceria and titania (CeOx/TiO2) generates active centers for CO2 adsorption and can facilitate the subsequent conversion of CO2 into methanol at low environmental pressures. This study underscores the importance of the localized electronic properties and structure of catalysts for achieving high alcohol selectivity from CO2 hydrogenation.
Why Does This Matter?
- Very small amounts of ceria are needed to stabilize the Au nanoparticles, significantly promoting their catalytic activity in producing methanol.
- This mechanism could be particularly effective in the remediation of greenhouse gas emissions from fossil fuel combustion, at the same time yielding commercially valuable byproducts.
What Are The Details?
CFN Capabilities: CFN’s Ambient Pressure Photoelectron Spectroscopy Endstation at X1A1 Beamline of NSLS (now at beamline CSX-2 of NSLS-II) was used to probe the role of CeOx and TiO2 in promoting the catalytic properties of Au.
Publication Reference
Low Pressure CO2 Hydrogenation to Methanol over Gold Nanoparticles Activated on a CeOx/TiO2 Interface
Xiaofang Yang,† Shyam Kattel,† Sanjaya D. Senanayake,† J. Anibal Boscoboinik,‡ Xiaowa Nie,§ Jesu´s Graciani,? Jose´ A. Rodriguez,† Ping Liu,† Darío J. Stacchiola,† and Jingguang G. Chen†,⊥
†Chemistry Department and ‡Center for Functional Nanomaterials (CFN), Brookhaven National Laboratory, Upton, New York 11973, United States
§Dalian University of Technology, Dalian 116024, Liaoning Province, China
?Department of Physical Chemistry, University of Seville, E-41012 Seville, Spain
⊥Department of Chemical Engineering, Columbia University, New York, New York 10027, United States
J. Am. Chem. Soc. 137, 10104−10107 (2015)
Acknowledgement of Support:
The work was sponsored under Contract No. DE-AC02-98CH10886 with the U.S. Department of Energy, Office of Science. This research used resources of the Center for Functional Nanomaterials and National Synchrotron Light Source, which are U.S. DOE Office of Science User Facilities at Brookhaven National Laboratory under Contract No. DESC0012704 and the National Energy Research Scientific Computing Center (NERSC) supported by the Office of Science of the US Department of Energy under Contract No. DE-AC02-05CH11231.
2015-6196 | INT/EXT | Newsroom









