Solving the Mystery of Fast Ion Transport in Batteries
April 30, 2020
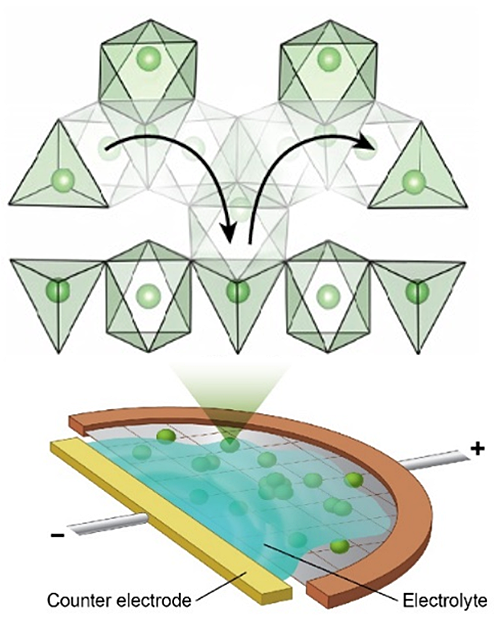
(Bottom) An electrochemically functional cell operated inside the transmission electron microscope tracks fast lithium-ion migration in a battery anode; which is enabled by distorted lithium polyhedra in the material (top).
What is the scientific achievement?
A collaborative team of scientists at Brookhaven Lab and UC Berkeley/Berkeley Lab used operando electron microscopy and computational modeling to observe and understand the underlying reasons that lithium titanate battery electrodes can support rapid charging and discharging. Spectroscopy measurements and supporting calculations show that this advantageous anode behavior is enabled by distorted lithium polyhedra in metastable intermediates during battery operation.
Why does this achievement matter?
This work provides a new understanding for designing fast-charging battery materials. A high-rate capability can be designed by accessing metastable structures above the ground state, which can have different kinetic mechanisms.
What are the details?
Fast-charging batteries typically use electrodes capable of accommodating lithium continuously by means of solid-solution transformation because they have few kinetic barriers apart from ionic diffusion. One exception is lithium titanate (Li4Ti5O12), an anode exhibiting extraordinary rate capability apparently inconsistent with its two-phase reaction and slow Li diffusion in both phases. Through real-time tracking of Li+ migration using operando electron energy-loss spectroscopy, we reveal that facile transport in Li4+xTi5O12 is enabled by kinetic pathways comprising distorted Li polyhedra in metastable intermediates along two-phase boundaries. Our work demonstrates that high-rate capability may be enabled by accessing the energy landscape above the ground state, which may have fundamentally different kinetic mechanisms from the ground-state macroscopic phases. This insight should present new opportunities in searching for high-rate electrode materials.
CFN Capabilities
The CFN Theory and Computation Facility was used to model the Li K-edge electron energy loss spectroscopy (EELS) spectra and to calculate the energy barriers for ionic transport.
Publication Reference
Zhang, D.-H. Seo, T. Chen, L. Wu, M. Topsakal, Y. Zhu, D. Lu, G. Ceder, F. Wang, Kinetic pathways of ionic transport in fast-charging lithium titanate, Science 367, 1030 (2020).
DOI: 10.1126/science.aax3520
https://science.sciencemag.org/content/367/6481/1030.abstract
https://doi.org/10.1126/science.aax3520
BNL news release: https://www.bnl.gov/newsroom/news.php?a=117060
LBL news release: https://newscenter.lbl.gov/2020/03/23/one-step-closer-to-a-fast-charging-battery/
OSTI Link: https://www.osti.gov/biblio/1601347-kinetic-pathways-ionic-transport-fast-charging-lithium-titanate
Acknowledgement of Support
This work was partially supported by the Laboratory Directed Research and Development program at the Brookhaven National Lab, under contract no. DE-SC0012704. This research used resources of the Center for Functional Nanomaterials, which is a U.S. Department of Energy (DOE) Office of Science Facility, and the Scientific Data and Computing Center, a component of the BNL Computational Science Initiative, at Brookhaven National Laboratory under Contract No. DE-SC0012704. This work was supported in part by the Assistant Secretary for Energy Efficiency and Renewable Energy, Vehicle Technologies Office, of the U.S. DOE under Contract No. DE-AC02-05CH11231, under the Advanced Battery Materials Research (BMR) Program and by the National Science Foundation Graduate Research Fellowship under Grant No. DGE 1106400. Efforts by F.W. involving writing and revision of the manuscript were supported by U.S. DOE Office of Energy Efficiency and Renewable Energy, Vehicle Technologies Office, Contract No. DE-SC0012704. Efforts on EELS data analysis by L.W. and Y.Z. were supported by the U.S. DOE, Office of Basic Energy Science, Division of Materials Science and Engineering under contract no. DE-SC0012704. T.C. thanks the National Science Foundation’s Extreme Science and Engineering Development Environment (XSEDE) supercomputer Stampede2, through allocation TG-DMR970008S, which is supported by National Science Foundation grant number ACI-1548562, for providing computing resources.
2020-17247 | INT/EXT | Newsroom









