Atomically Designed Imide-Based Nanoribbons Enable Efficient Organic Solar Cells
December 18, 2015
What Is The Scientific Achievement?
 enlarge
enlarge
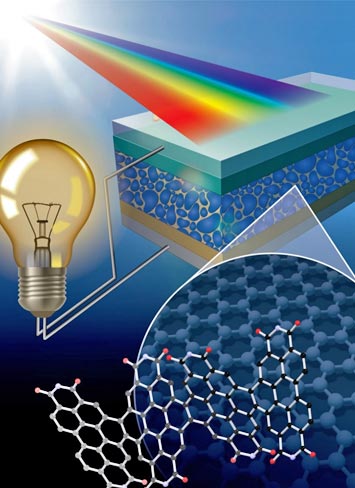
This study introduced a new class of graphene-based, highly efficient electron acceptor for solar cells. The solar cells described in this work possess power conversion efficiencies comparable to state-of-the-art polymer solar cells. The work identified here establishes a lower limit for what can be achieved; thus, a clear path for future development in carbon-based solar panels can be seen.
Low dimensional carbon nanomaterials are critical for the optimal performance of organic polymer-based solar cells (photovoltaics) as they can conduct photo-generated electrons with low losses. Among these materials, helical molecules have emerged as exceptional two-dimensional nanomaterials with excellent optical and electrical properties. In particular, helical imide-based nanoribbons (NRs), which are chemically synthesized, planar, helical molecules, combine efficient solar spectrum coverage, good electron mobility, and easy control over solar cell internal morphology, all critical for high organic solar cell performance. In this work, NRs incorporated into a photovoltaic system have yielded a 8.3% power conversion efficiency when used as the electron acceptor, a record high for organic polymer solar cells with non-fullerene-derived electron conductors.
Why Does This Matter?
These NRs have been proposed as low-cost alternatives to carbon fullerenes, which are the typical electron conducting carbon nanomaterials used in organic solar cells. Therefore, these results not only improve the cost advantages of organic photovoltaic (PV) technology over conventional inorganic solar cells but also provide groundbreaking guidelines for designing highly-efficient, electron conductors for high-performance organic PV and other optoelectronic devices.
What Are The Details?
CFN Capabilities: CFN’s Nanofabrication Facility was used for a component of device PV characterization. CFN’s Advanced Optical Spectroscopy & Microscopy Facility was used for the transient absorption spectroscopy measurement.
Publication Reference
Molecular helices as electron acceptors in high-performance bulk heterojunction solar cells
Yu Zhong1, M. Tuan Trinh1, Rongsheng Chen1,2, Geoffrey E. Purdum3, Petr P. Khlyabich3, Melda Sezen3, Seokjoon Oh1, Haiming Zhu1, Brandon Fowler1, Boyuan Zhang1, Wei Wang1, Chang-Yong Nam4, Matthew Y. Sfeir4, Charles T. Black4, Michael L. Steigerwald1, Yueh-Lin Loo3, Fay Ng1, X.-Y. Zhu1 & Colin Nuckolls1
1Department of Chemistry, Columbia University, 3000 Broadway, Havemeyer Hall, MC3130, New York, New York 10027, USA.
2School of Chemical Engineering and Technology, Wuhan University of Science and Technology, Wuhan 430081, China.
3Department of Chemical and Biological Engineering, Princeton University, Princeton, New Jersey 08544, USA.
4Center for Functional Nanomaterials, Brookhaven National Laboratory, Upton, New York, New York 11973, USA.
Nature Communications 6, 8242 (2015)
Acknowledgement of Support:
Primary support for this project was provided by the Chemical Sciences, Geosciences and Biosciences Division, Office of Basic Energy Sciences, US Department of Energy (DOE) under award number DE-FG02-01ER15264. X.Y.Z. acknowledges support by the US National Science Foundation grant DMR 1321405. This research used resources of the Center for Functional Nanomaterials, which is a US DOE Office of Science User Facility, at Brookhaven National Laboratory under contract number DE-SC0012704. Portions of this work were conducted at the Cornell High Energy Synchrotron Source (CHESS), which is supported by the National Science Foundation and the National Institutes of Health/National Institute of General Medical Sciences under NSF awards DMR-0936384 and DMR-1332208. G.E.P. acknowledges Government support under and awarded by DoD, Air Force Office of Scientific Research, National Defense Science and Engineering Graduate (NDSEG) Fellowship, 32 CFR 168a. We thank Columbia University’s Shared Materials Characterization Lab for use of the equipment essential to the research.
2015-6199 | INT/EXT | Newsroom









