Gas-Dependent Oxide Reduction Revealed at the Atomic Scale
January 28, 2026
 enlarge
enlarge
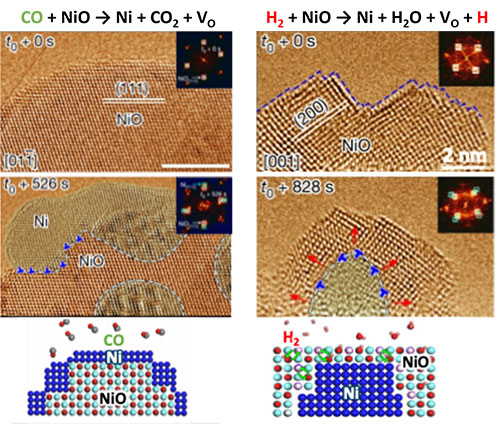
In-situ E-TEM images of NiO reduction at 400 °C under CO (left) and H2 (right), showing that, in CO, Ni islands form on NiO surface, leading to self-limiting surface metallization; while in H2, Ni islands form in bulk NiO, leading to bulk metallization.
Scientific Achievement
We show that CO and H2 reduce metal oxides through fundamentally different atomic pathways: CO quickly forms a surface metallic layer that blocks further reduction, while H2 enables reduction to proceed into the bulk by moving oxygen vacancies inward.
Significance and Impact
Oxide reduction is critical for metal production, catalysis, and energy technologies. By revealing gas-dependent reduction dynamics, this work provides a mechanistic foundation for metal production and catalyst design.
Research Details
Carbon monoxide (CO) and hydrogen (H2) are common reductants used in metal production, catalysis, and energy technologies. It was often thought that these reactions followed similar pathways, but a long-standing close collaboration among scientists from Binghamton University, Stony Brook University, Columbia University, and Brookhaven National Laboratory has found, for the first time, under direct atomic scale observation, that they follow fundamentally different reduction mechanisms.
Using the unique in situ environmental transmission electron microscopy (ETEM) at the Center for Functional Nanomaterials (CFN), a user facility at Department of Energy’s Brookhaven National Laboratory, the researchers compared the reduction dynamics of nickel oxide (NiO) under CO and H2 at atomic resolution under elevated temperatures. The atomic-scale movies reveal that CO reduction begins rapidly at the surface: metallic nickel forms quickly near the exterior, producing a thin “crust” that can block or slow continued reduction in the particle interior. As a result, CO reduction appears fast initially but can become self-limiting. In contrast, H2 reduction follows a very different pathway. For an extended initial period, metallic Ni is not observed; instead, the NiO shape changes which suggests vacancy movements. Metallic nickel then nucleates in the middle of the oxide, instead of on the surface. This allows the reduction to proceed into the bulk and continue advancing to fully reduce the oxide. Together, these in situ ETEM observations reveals two fundamentally different reduction pathways: CO quickly forms a surface metallic layer that blocks further reduction, while H2 enables reduction to proceed into the bulk by moving vacancies inward.
These in situ ETEM results are corroborated by Density functional theory (DFT) calculations, which indicate that protons from H2 promote oxygen-vacancy migration into the bulk, whereas under CO, oxygen vacancies remain largely confined near the surface.
To validate these findings on larger length scales, the team carried out complementary in situ measurements at the Quick X-ray Absorption and Scattering beamline at the National Synchrotron Light Source II (NSLS-II), another DOE user facility at Brookhaven. Using in situ X-ray diffraction, near-ambient-pressure X-ray photoelectron spectroscopy, and residual gas analysis, they confirmed gas-dependent differences in NiO reduction behavior at the bulk scale.
This atomic scale understanding of the reduction process provides guidance for metallurgical processes and catalyst design. By showing that the choice of reductant gas directly influences where and how metallic phases form, this work enables more precise control over reduction depth, rate, and resulting microstructure.
Publication Reference
Xiaobo Chen, Jianyu Wang, Shyam Bharatkumar Patel, Shuonan Ye, Yupeng Wu, Zhikang Zhou, Linna Qiao, Yuxi Wang, Nebojsa Marinkovic, Meng Li, Sooyeon Hwang, Dmitri N. Zakharov, Lu Ma, Qin Wu, Jorge Anibal Boscoboinik, Judith C. Yang,Guangwen Zhou, Atomic dynamics of gas-dependent oxide reducibility. Nature 644, 927–932 (2025). https://doi.org/10.1038/s41586-025-09394-0
DOI: https://www.nature.com/articles/s41586-025-09394-0
Press Release: New research could lead to greener, faster metal production
Acknowledgment of Support
This work was supported by the U.S. National Science Foundation (Grant No. DMR 2303712). The computational modelling work was supported by the U.S. Department of Energy, Office of Basic Energy Sciences, Division of Materials Sciences and Engineering (Award No. DE-SC0001135). This research used the Electron Microscopy, Proximal Probes, and Theory and Computation resources of the Center for Functional Nanomaterials and beamline 7-BM (QAS) at the National Synchrotron Light Source II, which are U.S. DOE Office of Science User Facilities, at Brookhaven National Laboratory under Contract No. DE-SC0012704. Work at the beamline was supported in part by the Synchrotron Catalysis Consortium, U.S. Department of Energy (Grant No. DE-SC0012335).
2026-22837 | INT/EXT | Newsroom









