Optimization of Lead Sulfide Quantum Dot Synthesis for Monodispersity
August 12, 2014
 enlarge
enlarge
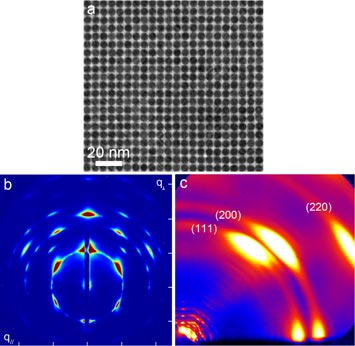
a) Transmission electron micrograph of a QD superlattice, highlighting the narrow size dispersity b) Grazing-incidence small-angle X-ray scattering pattern of a large area (1 mm2) QD film arranged in a body-centered cubic lattice c) Wide-angle X-ray scattering pattern from the same film, showing that the facets of the individual QDs also are aligned within the superlattice.
What Is The Scientific Achievement?
Semiconductor quantum dots (QDs) are nanoscale semiconductors that exhibit size dependent physical properties. For example, the color (wavelength) of light that they absorb changes dramatically as the diameter decreases. This phenomenon is due to a concept called quantum confinement and is achieved when the physical size of the QD is smaller than the size of the orbit of the electrons (represented by the electron wavefunction) in bulk forms of the material. In this research project, a new method was developed to synthesize highly monodisperse lead sulfide quantum dots (PbS QDs) over a wide range of sizes. Historically, PbS QDs have been synthesized with an acceptable size dispersity of ~10 %. In this new study, PbS QDs were synthesized with band gaps ranging from 0.7 eV to 1.25 eV (4–8 nm in diameter) with a size dispersity < 3%, which is 3 times better than the typical monodisperse system. As a result of their monodispersity, the QDs readily self-assemble into highly ordered superlattice structures.
Why Does This Matter?
The resultant nanoparticles are so highly monodisperse that they readily self-assemble into large-area ordered arrays (superlattices), as can be seen in electron microscopy, and in x-ray scattering. Well-ordered superlattices of QDs possess enhanced electrical properties within the film, giving rise to potentially coherent charge transport phenomena within the QD structures.
What Are The Details?
- CFN Capabilities: CFN’s Advanced UV and X-ray Probes Facility (X9 beamline at NSLS) was critical for measuring the macro-scale self-assembly of the QD superlattices, probing the nanoscale packing and the orientation of particles within the lattice.
Publication Reference
Monodisperse, Air-Stable PbS Nanocrystals via Precursor Stoichiometry Control
Mark C. Weidman, Megan E. Beck, Rachel S. Hoffman, Ferry Prins, and William A. Tisdale
Department of Chemical Engineering, Massachusetts Institute of Technology, Cambridge, Massachusetts 02139, United States
ACS Nano 8, 6363-6371 (2014)
Acknowledgment of Support
This work was primarily supported by the U.S. Department of Energy, Office of Basic Energy Sciences, under Award Number DE-SC0010538. M.C.W. was supported by a National Science Foundation Graduate Research Fellowship under Grant No. 1122374. TEM, XRD, XPS, and SAXS measurements were performed in the MRSEC Shared Experimental Facilities at MIT, supported by the National Science Foundation under award number DMR-08-19762. GISAXS and WAXS measurements were performed at the Center for Functional Nanomaterials and the National Synchrotron Light Source, Brookhaven National Laboratory, which is supported by the U.S. Department of Energy, Office of Basic Energy Sciences, under Contract No. DE-AC02-98CH10886.
2014-5120 | INT/EXT | Newsroom











